+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7dfz | ||||||
|---|---|---|---|---|---|---|---|
| Title | Cryo_EM structure of delta N-NPC1L1-EZE | ||||||
 Components Components | NPC1-like intracellular cholesterol transporter 1 | ||||||
 Keywords Keywords | STRUCTURAL PROTEIN/INHIBITOR / Ezetimibe / STRUCTURAL PROTEIN-INHIBITOR Complex | ||||||
| Function / homology |  Function and homology information Function and homology informationvitamin transport / cholesterol import / lipid transporter activity / response to muscle activity / heterocyclic compound binding / brush border membrane / apical plasma membrane / response to xenobiotic stimulus Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.58 Å | ||||||
 Authors Authors | Hu, M. / Sun, S. | ||||||
 Citation Citation |  Journal: Sci Adv / Year: 2021 Journal: Sci Adv / Year: 2021Title: Structural insights into the mechanism of human NPC1L1-mediated cholesterol uptake. Authors: Miaoqing Hu / Fan Yang / Yawen Huang / Xin You / Desheng Liu / Shan Sun / Sen-Fang Sui /  Abstract: Niemann-Pick C1-like 1 (NPC1L1) protein plays a central role in the intestinal cholesterol absorption and is the target of a drug, ezetimibe, which inhibits NPC1L1 to reduce cholesterol absorption. ...Niemann-Pick C1-like 1 (NPC1L1) protein plays a central role in the intestinal cholesterol absorption and is the target of a drug, ezetimibe, which inhibits NPC1L1 to reduce cholesterol absorption. Here, we present cryo-electron microscopy structures of human NPC1L1 in apo state, cholesterol-enriched state, and ezetimibe-bound state to reveal molecular details of NPC1L1-mediated cholesterol uptake and ezetimibe inhibition. Comparison of these structures reveals that the sterol-sensing domain (SSD) could respond to the cholesterol level alteration by binding different number of cholesterol molecules. Upon increasing cholesterol level, SSD binds more cholesterol molecules, which, in turn, triggers the formation of a stable structural cluster in SSD, while binding of ezetimibe causes the deformation of the SSD and destroys the structural cluster, leading to the inhibition of NPC1L1 function. These results provide insights into mechanisms of NPC1L1 function and ezetimibe action and are of great significance for the development of new cholesterol absorption inhibitors. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7dfz.cif.gz 7dfz.cif.gz | 178.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7dfz.ent.gz pdb7dfz.ent.gz | 145.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7dfz.json.gz 7dfz.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  7dfz_validation.pdf.gz 7dfz_validation.pdf.gz | 1.3 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  7dfz_full_validation.pdf.gz 7dfz_full_validation.pdf.gz | 1.3 MB | Display | |
| Data in XML |  7dfz_validation.xml.gz 7dfz_validation.xml.gz | 35.9 KB | Display | |
| Data in CIF |  7dfz_validation.cif.gz 7dfz_validation.cif.gz | 51.7 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/df/7dfz https://data.pdbj.org/pub/pdb/validation_reports/df/7dfz ftp://data.pdbj.org/pub/pdb/validation_reports/df/7dfz ftp://data.pdbj.org/pub/pdb/validation_reports/df/7dfz | HTTPS FTP |
-Related structure data
| Related structure data |  30668MC  7df8C  7dfwC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 139612.125 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: NPC1L1 / Cell line (production host): HEK293 / Production host: Homo sapiens (human) / Gene: NPC1L1 / Cell line (production host): HEK293 / Production host:  Homo sapiens (human) / References: UniProt: A0A0C4DFX6 Homo sapiens (human) / References: UniProt: A0A0C4DFX6 |
|---|
-Sugars , 4 types, 10 molecules 
| #2: Polysaccharide | Source method: isolated from a genetically manipulated source #3: Polysaccharide | 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-3)-2-acetamido-2-deoxy-beta-D-glucopyranose | Source method: isolated from a genetically manipulated source #4: Polysaccharide | Source method: isolated from a genetically manipulated source #5: Sugar | ChemComp-NAG / |
|---|
-Non-polymers , 2 types, 5 molecules 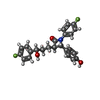


| #6: Chemical | ChemComp-H56 / ( |
|---|---|
| #7: Chemical | ChemComp-CLR / |
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: delta N-hNPC1L1-EZE / Type: COMPLEX / Entity ID: #1 / Source: RECOMBINANT |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:  Homo sapiens (human) / Cell: HEK293 Homo sapiens (human) / Cell: HEK293 |
| Buffer solution | pH: 7.4 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER |
| Electron lens | Mode: OTHER |
| Image recording | Electron dose: 50 e/Å2 / Film or detector model: GATAN K2 SUMMIT (4k x 4k) |
- Processing
Processing
| CTF correction | Type: NONE |
|---|---|
| 3D reconstruction | Resolution: 3.58 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 2204129 / Symmetry type: POINT |
 Movie
Movie Controller
Controller
















 PDBj
PDBj









