[English] 日本語
 Yorodumi
Yorodumi- PDB-7a1z: Crystal structure of human protein kinase CK2alpha' (CSNK2A2 gene... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7a1z | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of human protein kinase CK2alpha' (CSNK2A2 gene product) in complex with the ATP-competitive inhibitor 6-bromo-5-chloro-1H-triazolo[4,5-b]pyridine | ||||||
 Components Components | Casein kinase II subunit alpha' | ||||||
 Keywords Keywords | TRANSFERASE / protein kinase CK2 / casein kinase 2 / ATP-competitive inhibitor | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of mitophagy / regulation of chromosome separation / WNT mediated activation of DVL / Condensation of Prometaphase Chromosomes / protein kinase CK2 complex / : / Phosphorylation and nuclear translocation of the CRY:PER:kinase complex / Regulation of CDH1 posttranslational processing and trafficking to plasma membrane / Receptor Mediated Mitophagy / Synthesis of PC ...regulation of mitophagy / regulation of chromosome separation / WNT mediated activation of DVL / Condensation of Prometaphase Chromosomes / protein kinase CK2 complex / : / Phosphorylation and nuclear translocation of the CRY:PER:kinase complex / Regulation of CDH1 posttranslational processing and trafficking to plasma membrane / Receptor Mediated Mitophagy / Synthesis of PC / RUNX1 interacts with co-factors whose precise effect on RUNX1 targets is not known / Maturation of hRSV A proteins / negative regulation of apoptotic signaling pathway / negative regulation of proteasomal ubiquitin-dependent protein catabolic process / liver regeneration / acrosomal vesicle / Signal transduction by L1 / cerebral cortex development / Wnt signaling pathway / Regulation of PTEN stability and activity / KEAP1-NFE2L2 pathway / double-strand break repair / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / spermatogenesis / Regulation of TP53 Activity through Phosphorylation / non-specific serine/threonine protein kinase / protein serine kinase activity / protein serine/threonine kinase activity / apoptotic process / DNA damage response / chromatin / nucleoplasm / ATP binding / nucleus / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / AB INITIO PHASING / Resolution: 1.024 Å SYNCHROTRON / AB INITIO PHASING / Resolution: 1.024 Å | ||||||
 Authors Authors | Niefind, K. / Lindenblatt, D. / Toelzer, C. / Bretner, M. / Chojnacki, K. / Wielechowska, M. / Winska, P. | ||||||
| Funding support |  Germany, 1items Germany, 1items
| ||||||
 Citation Citation |  Journal: Bioorg.Chem. / Year: 2021 Journal: Bioorg.Chem. / Year: 2021Title: Synthesis, biological properties and structural study of new halogenated azolo[4,5-b]pyridines as inhibitors of CK2 kinase. Authors: Chojnacki, K. / Lindenblatt, D. / Winska, P. / Wielechowska, M. / Toelzer, C. / Niefind, K. / Bretner, M. #1:  Journal: Bioorg. Chem. / Year: 2018 Journal: Bioorg. Chem. / Year: 2018Title: Biological properties and structural study of new aminoalkyl derivatives of benzimidazole and benzotriazole, dual inhibitors of CK2 and PIM1 kinases. Authors: Chojnacki, K. / Winska, P. / Wielechowska, M. / Lukowska-Chojnacka, E. / Toelzer, C. / Niefind, K. / Bretner, M. #2:  Journal: J. Med. Chem. / Year: 2020 Journal: J. Med. Chem. / Year: 2020Title: Structural and Mechanistic Basis of the Inhibitory Potency of Selected 2-Aminothiazole Compounds on Protein Kinase CK2. Authors: Lindenblatt, D. / Nickelsen, A. / Applegate, V.M. / Jose, J. / Niefind, K. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7a1z.cif.gz 7a1z.cif.gz | 280.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7a1z.ent.gz pdb7a1z.ent.gz | 190.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7a1z.json.gz 7a1z.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/a1/7a1z https://data.pdbj.org/pub/pdb/validation_reports/a1/7a1z ftp://data.pdbj.org/pub/pdb/validation_reports/a1/7a1z ftp://data.pdbj.org/pub/pdb/validation_reports/a1/7a1z | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7a1bC  7a22C  7a2hC  7a49C  7a4bC  7a4cC C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 42879.867 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CSNK2A2, CK2A2 / Production host: Homo sapiens (human) / Gene: CSNK2A2, CK2A2 / Production host:  References: UniProt: P19784, non-specific serine/threonine protein kinase | ||||
|---|---|---|---|---|---|
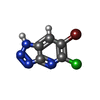
| #2: Chemical | ChemComp-QWN / | ||||
| #3: Chemical | ChemComp-CL / | ||||
| #4: Chemical | ChemComp-EDO / #5: Water | ChemComp-HOH / | Has ligand of interest | Y | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.62 Å3/Da / Density % sol: 53.06 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop Details: Reservoir composition: 28 % (w/v) PEG6000, 0.9 M LiCl, 0.1 M, Tris/HCl, pH 8.5; drop composition prior to equilibration: 0.01 ml reservoir solution + 0.02 ml CK2alpha' (mutant Cys336Ser) ...Details: Reservoir composition: 28 % (w/v) PEG6000, 0.9 M LiCl, 0.1 M, Tris/HCl, pH 8.5; drop composition prior to equilibration: 0.01 ml reservoir solution + 0.02 ml CK2alpha' (mutant Cys336Ser)/inhibitor MB002 mixture (0.180 ml 6 mg/ml CK2alpha'Cys336Ser, 0.5 M NaCl, 25 mM Tris/HCl, pH 8.5, mixed and pre-equilibrated with 0.02 ml 10 mM MB002 in dimethyl sulfoxide); the initial inhibitor MB002 was replaced by the inhibitor 6-bromo-5-chloro-1H-triazolo[4,5-b]pyridine by extensive crystal soaking. |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID23-2 / Wavelength: 0.87313 Å / Beamline: ID23-2 / Wavelength: 0.87313 Å |
| Detector | Type: DECTRIS PILATUS 2M / Detector: PIXEL / Date: Nov 14, 2018 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.87313 Å / Relative weight: 1 |
| Reflection | Resolution: 1.024→46.434 Å / Num. obs: 155664 / % possible obs: 77.7 % / Redundancy: 9 % / Biso Wilson estimate: 10.52 Å2 / CC1/2: 0.999 / Rmerge(I) obs: 0.088 / Rpim(I) all: 0.03 / Rrim(I) all: 0.093 / Rsym value: 0.088 / Net I/σ(I): 12 |
| Reflection shell | Resolution: 1.024→1.086 Å / Rmerge(I) obs: 1.38 / Mean I/σ(I) obs: 1.6 / Num. unique obs: 7786 / CC1/2: 0.5 / Rpim(I) all: 0.596 / Rrim(I) all: 1.513 / Rsym value: 1.38 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure: AB INITIO PHASING / Resolution: 1.024→46.43 Å / SU ML: 0.0778 / Cross valid method: FREE R-VALUE / σ(F): 1.96 / Phase error: 17.8544 Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 17.21 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.024→46.43 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller





















 PDBj
PDBj