+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-4654 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
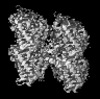
| Title | HsCKK (human CAMSAP1) decorated 14pf taxol-GDP microtubule | |||||||||
 Map data Map data | HsCKK (Human CKK) decorated 14pf taxol-GDP microtubule (symmetrised asymmetric unit) | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Microtubule CAMSAP Calmodulin-regulated spectrum-associated proteins CKK Cryo-EM Cryo-Electron Microscopy / STRUCTURAL PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationmicrotubule minus-end binding / odontoblast differentiation / Post-chaperonin tubulin folding pathway / Microtubule-dependent trafficking of connexons from Golgi to the plasma membrane / Cilium Assembly / Carboxyterminal post-translational modifications of tubulin / Sealing of the nuclear envelope (NE) by ESCRT-III / Intraflagellar transport / cytoskeleton-dependent intracellular transport / Formation of tubulin folding intermediates by CCT/TriC ...microtubule minus-end binding / odontoblast differentiation / Post-chaperonin tubulin folding pathway / Microtubule-dependent trafficking of connexons from Golgi to the plasma membrane / Cilium Assembly / Carboxyterminal post-translational modifications of tubulin / Sealing of the nuclear envelope (NE) by ESCRT-III / Intraflagellar transport / cytoskeleton-dependent intracellular transport / Formation of tubulin folding intermediates by CCT/TriC / Gap junction assembly / COPI-independent Golgi-to-ER retrograde traffic / regulation of cell morphogenesis / Kinesins / Hedgehog 'off' state / Assembly and cell surface presentation of NMDA receptors / GTPase activating protein binding / microtubule organizing center / COPI-dependent Golgi-to-ER retrograde traffic / natural killer cell mediated cytotoxicity / spectrin binding / intercellular bridge / regulation of synapse organization / nuclear envelope lumen / MHC class I protein binding / cytoplasmic microtubule / regulation of microtubule polymerization / Recycling pathway of L1 / microtubule-based process / RHOH GTPase cycle / RHO GTPases activate IQGAPs / spindle assembly / cellular response to interleukin-4 / COPI-mediated anterograde transport / Activation of AMPK downstream of NMDARs / Mitotic Prometaphase / EML4 and NUDC in mitotic spindle formation / Loss of Nlp from mitotic centrosomes / Loss of proteins required for interphase microtubule organization from the centrosome / Recruitment of mitotic centrosome proteins and complexes / cytoskeleton organization / Recruitment of NuMA to mitotic centrosomes / Anchoring of the basal body to the plasma membrane / HSP90 chaperone cycle for steroid hormone receptors (SHR) in the presence of ligand / Resolution of Sister Chromatid Cohesion / MHC class II antigen presentation / AURKA Activation by TPX2 / Translocation of SLC2A4 (GLUT4) to the plasma membrane / RHO GTPases Activate Formins / PKR-mediated signaling / mitotic spindle / structural constituent of cytoskeleton / microtubule cytoskeleton organization / HCMV Early Events / Aggrephagy / cytoplasmic ribonucleoprotein granule / Separation of Sister Chromatids / The role of GTSE1 in G2/M progression after G2 checkpoint / azurophil granule lumen / microtubule cytoskeleton / Regulation of PLK1 Activity at G2/M Transition / double-stranded RNA binding / neuron projection development / Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement / mitotic cell cycle / cell body / microtubule binding / microtubule / Potential therapeutics for SARS / cytoskeleton / calmodulin binding / membrane raft / protein domain specific binding / cell division / GTPase activity / ubiquitin protein ligase binding / Neutrophil degranulation / GTP binding / protein-containing complex binding / structural molecule activity / protein-containing complex / extracellular exosome / extracellular region / nucleus / metal ion binding / cytosol / cytoplasm Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.8 Å | |||||||||
 Authors Authors | Atherton JM / Luo Y | |||||||||
| Funding support |  United Kingdom, United Kingdom,  Switzerland, 2 items Switzerland, 2 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2019 Journal: Nat Commun / Year: 2019Title: Structural determinants of microtubule minus end preference in CAMSAP CKK domains. Authors: Joseph Atherton / Yanzhang Luo / Shengqi Xiang / Chao Yang / Ankit Rai / Kai Jiang / Marcel Stangier / Annapurna Vemu / Alexander D Cook / Su Wang / Antonina Roll-Mecak / Michel O Steinmetz ...Authors: Joseph Atherton / Yanzhang Luo / Shengqi Xiang / Chao Yang / Ankit Rai / Kai Jiang / Marcel Stangier / Annapurna Vemu / Alexander D Cook / Su Wang / Antonina Roll-Mecak / Michel O Steinmetz / Anna Akhmanova / Marc Baldus / Carolyn A Moores /      Abstract: CAMSAP/Patronins regulate microtubule minus-end dynamics. Their end specificity is mediated by their CKK domains, which we proposed recognise specific tubulin conformations found at minus ends. To ...CAMSAP/Patronins regulate microtubule minus-end dynamics. Their end specificity is mediated by their CKK domains, which we proposed recognise specific tubulin conformations found at minus ends. To critically test this idea, we compared the human CAMSAP1 CKK domain (HsCKK) with a CKK domain from Naegleria gruberi (NgCKK), which lacks minus-end specificity. Here we report near-atomic cryo-electron microscopy structures of HsCKK- and NgCKK-microtubule complexes, which show that these CKK domains share the same protein fold, bind at the intradimer interprotofilament tubulin junction, but exhibit different footprints on microtubules. NMR experiments show that both HsCKK and NgCKK are remarkably rigid. However, whereas NgCKK binding does not alter the microtubule architecture, HsCKK remodels its microtubule interaction site and changes the underlying polymer structure because the tubulin lattice conformation is not optimal for its binding. Thus, in contrast to many MAPs, the HsCKK domain can differentiate subtly specific tubulin conformations to enable microtubule minus-end recognition. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_4654.map.gz emd_4654.map.gz | 641.5 KB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-4654-v30.xml emd-4654-v30.xml emd-4654.xml emd-4654.xml | 18.2 KB 18.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_4654.png emd_4654.png | 208.9 KB | ||
| Filedesc metadata |  emd-4654.cif.gz emd-4654.cif.gz | 6.9 KB | ||
| Others |  emd_4654_additional.map.gz emd_4654_additional.map.gz | 172.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-4654 http://ftp.pdbj.org/pub/emdb/structures/EMD-4654 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4654 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4654 | HTTPS FTP |
-Validation report
| Summary document |  emd_4654_validation.pdf.gz emd_4654_validation.pdf.gz | 225.9 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_4654_full_validation.pdf.gz emd_4654_full_validation.pdf.gz | 225 KB | Display | |
| Data in XML |  emd_4654_validation.xml.gz emd_4654_validation.xml.gz | 4.2 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4654 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4654 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4654 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4654 | HTTPS FTP |
-Related structure data
| Related structure data |  6qvjMC  4643C  4644C  4650C  6qusC  6quyC  6qveC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_4654.map.gz / Format: CCP4 / Size: 307.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_4654.map.gz / Format: CCP4 / Size: 307.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | HsCKK (Human CKK) decorated 14pf taxol-GDP microtubule (symmetrised asymmetric unit) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. generated in cubic-lattice coordinate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.39 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: HsCKK (Human CKK) decorated 14pf taxol-GDP microtubule (C1...
| File | emd_4654_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | HsCKK (Human CKK) decorated 14pf taxol-GDP microtubule (C1 reconstruction) | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : HsCKK (human CAMSAP1) decorated 14pf taxol-GDP microtubule
+Supramolecule #1: HsCKK (human CAMSAP1) decorated 14pf taxol-GDP microtubule
+Supramolecule #2: CAMSAP1
+Supramolecule #3: tubilin
+Macromolecule #1: Calmodulin-regulated spectrin-associated protein 1
+Macromolecule #2: Tubulin alpha-1B chain
+Macromolecule #3: Tubulin beta chain
+Macromolecule #4: GUANOSINE-5'-TRIPHOSPHATE
+Macromolecule #5: MAGNESIUM ION
+Macromolecule #6: GUANOSINE-5'-DIPHOSPHATE
+Macromolecule #7: TAXOL
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 6.8 / Details: BRB20 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI POLARA 300 |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 42.0 e/Å2 / Details: dose weighted images used in final reconstructions |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.8 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 33419 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: RELION |
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: OTHER |
|---|---|
| Output model |  PDB-6qvj: |
 Movie
Movie Controller
Controller








































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)