[日本語] English
 万見
万見- EMDB-4590: Leishmania tarentolae proteasome 20S subunit complexed with GSK3494245 -
+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-4590 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Leishmania tarentolae proteasome 20S subunit complexed with GSK3494245 | |||||||||
 マップデータ マップデータ | This is the sharpened EM map which was used for building the protein coordinates | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | Proteasome 20S subunit / hydrolase | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報proteasome core complex / proteasome endopeptidase complex / proteasome core complex, beta-subunit complex / threonine-type endopeptidase activity / proteasome core complex, alpha-subunit complex / proteasomal protein catabolic process / proteolysis involved in protein catabolic process / ubiquitin-dependent protein catabolic process / proteasome-mediated ubiquitin-dependent protein catabolic process / hydrolase activity ...proteasome core complex / proteasome endopeptidase complex / proteasome core complex, beta-subunit complex / threonine-type endopeptidase activity / proteasome core complex, alpha-subunit complex / proteasomal protein catabolic process / proteolysis involved in protein catabolic process / ubiquitin-dependent protein catabolic process / proteasome-mediated ubiquitin-dependent protein catabolic process / hydrolase activity / nucleus / cytoplasm 類似検索 - 分子機能 | |||||||||
| 生物種 |  Leishmania tarentolae (真核生物) Leishmania tarentolae (真核生物) | |||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 2.8 Å | |||||||||
 データ登録者 データ登録者 | Rowland P / Goswami P | |||||||||
 引用 引用 |  ジャーナル: Proc Natl Acad Sci U S A / 年: 2019 ジャーナル: Proc Natl Acad Sci U S A / 年: 2019タイトル: Preclinical candidate for the treatment of visceral leishmaniasis that acts through proteasome inhibition. 著者: Susan Wyllie / Stephen Brand / Michael Thomas / Manu De Rycker / Chun-Wa Chung / Imanol Pena / Ryan P Bingham / Juan A Bueren-Calabuig / Juan Cantizani / David Cebrian / Peter D Craggs / Liam ...著者: Susan Wyllie / Stephen Brand / Michael Thomas / Manu De Rycker / Chun-Wa Chung / Imanol Pena / Ryan P Bingham / Juan A Bueren-Calabuig / Juan Cantizani / David Cebrian / Peter D Craggs / Liam Ferguson / Panchali Goswami / Judith Hobrath / Jonathan Howe / Laura Jeacock / Eun-Jung Ko / Justyna Korczynska / Lorna MacLean / Sujatha Manthri / Maria S Martinez / Lydia Mata-Cantero / Sonia Moniz / Andrea Nühs / Maria Osuna-Cabello / Erika Pinto / Jennifer Riley / Sharon Robinson / Paul Rowland / Frederick R C Simeons / Yoko Shishikura / Daniel Spinks / Laste Stojanovski / John Thomas / Stephen Thompson / Elisabet Viayna Gaza / Richard J Wall / Fabio Zuccotto / David Horn / Michael A J Ferguson / Alan H Fairlamb / Jose M Fiandor / Julio Martin / David W Gray / Timothy J Miles / Ian H Gilbert / Kevin D Read / Maria Marco / Paul G Wyatt /   要旨: Visceral leishmaniasis (VL), caused by the protozoan parasites and , is one of the major parasitic diseases worldwide. There is an urgent need for new drugs to treat VL, because current therapies ...Visceral leishmaniasis (VL), caused by the protozoan parasites and , is one of the major parasitic diseases worldwide. There is an urgent need for new drugs to treat VL, because current therapies are unfit for purpose in a resource-poor setting. Here, we describe the development of a preclinical drug candidate, GSK3494245/DDD01305143/compound 8, with potential to treat this neglected tropical disease. The compound series was discovered by repurposing hits from a screen against the related parasite Subsequent optimization of the chemical series resulted in the development of a potent cidal compound with activity against a range of clinically relevant and isolates. Compound 8 demonstrates promising pharmacokinetic properties and impressive in vivo efficacy in our mouse model of infection comparable with those of the current oral antileishmanial miltefosine. Detailed mode of action studies confirm that this compound acts principally by inhibition of the chymotrypsin-like activity catalyzed by the β5 subunit of the proteasome. High-resolution cryo-EM structures of apo and compound 8-bound 20S proteasome reveal a previously undiscovered inhibitor site that lies between the β4 and β5 proteasome subunits. This induced pocket exploits β4 residues that are divergent between humans and kinetoplastid parasites and is consistent with all of our experimental and mutagenesis data. As a result of these comprehensive studies and due to a favorable developability and safety profile, compound 8 is being advanced toward human clinical trials. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_4590.map.gz emd_4590.map.gz | 9.5 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-4590-v30.xml emd-4590-v30.xml emd-4590.xml emd-4590.xml | 26 KB 26 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_4590.png emd_4590.png | 177.2 KB | ||
| Filedesc metadata |  emd-4590.cif.gz emd-4590.cif.gz | 8.8 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-4590 http://ftp.pdbj.org/pub/emdb/structures/EMD-4590 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4590 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4590 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_4590_validation.pdf.gz emd_4590_validation.pdf.gz | 238.5 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_4590_full_validation.pdf.gz emd_4590_full_validation.pdf.gz | 237.6 KB | 表示 | |
| XML形式データ |  emd_4590_validation.xml.gz emd_4590_validation.xml.gz | 6.5 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4590 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4590 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4590 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4590 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_4590.map.gz / 形式: CCP4 / 大きさ: 103 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_4590.map.gz / 形式: CCP4 / 大きさ: 103 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | This is the sharpened EM map which was used for building the protein coordinates | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
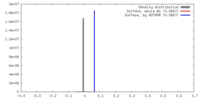
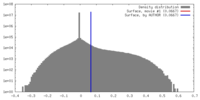
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
- 試料の構成要素
試料の構成要素
+全体 : Proteasome 20S subunit
+超分子 #1: Proteasome 20S subunit
+分子 #1: Proteasome alpha1 chain
+分子 #2: Proteasome alpha2 chain
+分子 #3: Proteasome alpha3 chain
+分子 #4: Proteasome alpha4 chain
+分子 #5: Proteasome alpha5 chain
+分子 #6: Proteasome alpha6 chain
+分子 #7: Proteasome alpha7 chain
+分子 #8: Proteasome beta1 chain
+分子 #9: Proteasome beta2 chain
+分子 #10: Proteasome beta3 chain
+分子 #11: Proteasome beta4 chain
+分子 #12: Proteasome beta5 chain
+分子 #13: Proteasome beta6 chain
+分子 #14: Proteasome beta7 chain
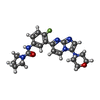
+分子 #15: ~{N}-[4-fluoranyl-3-(3-morpholin-4-ylimidazo[1,2-a]pyrimidin-7-yl...
+分子 #16: water
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.5 |
|---|---|
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: FEI FALCON III (4k x 4k) 検出モード: COUNTING / 平均電子線量: 30.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 初期モデル | モデルのタイプ: PDB ENTRY |
|---|---|
| 最終 再構成 | 想定した対称性 - 点群: C1 (非対称) / 解像度のタイプ: BY AUTHOR / 解像度: 2.8 Å / 解像度の算出法: FSC 0.143 CUT-OFF / ソフトウェア - 名称: RELION (ver. 2.1) / 使用した粒子像数: 182775 |
| 初期 角度割当 | タイプ: MAXIMUM LIKELIHOOD |
| 最終 角度割当 | タイプ: MAXIMUM LIKELIHOOD |
 ムービー
ムービー コントローラー
コントローラー



















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)