+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3ftu | ||||||
|---|---|---|---|---|---|---|---|
| Title | Leukotriene A4 hydrolase in complex with dihydroresveratrol | ||||||
 Components Components | Leukotriene A-4 hydrolase | ||||||
 Keywords Keywords | HYDROLASE / Leukotriene A4 Hydrolase / LTA4H / Fragment crystallography / Fragments of Life / FOL / Resveratrol / Alternative splicing / Cytoplasm / Leukotriene biosynthesis / Metal-binding / Metalloprotease / Multifunctional enzyme / Polymorphism / Protease / Zinc | ||||||
| Function / homology |  Function and homology information Function and homology informationleukotriene-A4 hydrolase / leukotriene-A4 hydrolase activity / tripeptide aminopeptidase activity / tripeptide aminopeptidase / Biosynthesis of protectins / Biosynthesis of aspirin-triggered D-series resolvins / Biosynthesis of E-series 18(R)-resolvins / Biosynthesis of D-series resolvins / Biosynthesis of E-series 18(S)-resolvins / Synthesis of Leukotrienes (LT) and Eoxins (EX) ...leukotriene-A4 hydrolase / leukotriene-A4 hydrolase activity / tripeptide aminopeptidase activity / tripeptide aminopeptidase / Biosynthesis of protectins / Biosynthesis of aspirin-triggered D-series resolvins / Biosynthesis of E-series 18(R)-resolvins / Biosynthesis of D-series resolvins / Biosynthesis of E-series 18(S)-resolvins / Synthesis of Leukotrienes (LT) and Eoxins (EX) / epoxide hydrolase activity / leukotriene biosynthetic process / response to zinc ion / peptide catabolic process / metalloaminopeptidase activity / type I pneumocyte differentiation / aminopeptidase activity / lipid metabolic process / response to peptide hormone / tertiary granule lumen / peptidase activity / ficolin-1-rich granule lumen / Neutrophil degranulation / proteolysis / RNA binding / extracellular exosome / extracellular region / zinc ion binding / nucleoplasm / nucleus / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.9 Å MOLECULAR REPLACEMENT / Resolution: 1.9 Å | ||||||
 Authors Authors | Davies, D.R. | ||||||
 Citation Citation |  Journal: J.Med.Chem. / Year: 2009 Journal: J.Med.Chem. / Year: 2009Title: Discovery of leukotriene A4 hydrolase inhibitors using metabolomics biased fragment crystallography. Authors: Davies, D.R. / Mamat, B. / Magnusson, O.T. / Christensen, J. / Haraldsson, M.H. / Mishra, R. / Pease, B. / Hansen, E. / Singh, J. / Zembower, D. / Kim, H. / Kiselyov, A.S. / Burgin, A.B. / ...Authors: Davies, D.R. / Mamat, B. / Magnusson, O.T. / Christensen, J. / Haraldsson, M.H. / Mishra, R. / Pease, B. / Hansen, E. / Singh, J. / Zembower, D. / Kim, H. / Kiselyov, A.S. / Burgin, A.B. / Gurney, M.E. / Stewart, L.J. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3ftu.cif.gz 3ftu.cif.gz | 142.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3ftu.ent.gz pdb3ftu.ent.gz | 108.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3ftu.json.gz 3ftu.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ft/3ftu https://data.pdbj.org/pub/pdb/validation_reports/ft/3ftu ftp://data.pdbj.org/pub/pdb/validation_reports/ft/3ftu ftp://data.pdbj.org/pub/pdb/validation_reports/ft/3ftu | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3ftsC  3ftvC  3ftwC  3ftxC  3ftyC  3fu0C  3fu3C  3fu5C  3fu6C  3fudC  3fueC  3fufC  3fuhC  3fuiC  3fujC  3fukC  3fumC  3funC  3fh7S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 69363.969 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: LTA4H, LTA4 / Production host: Homo sapiens (human) / Gene: LTA4H, LTA4 / Production host:  |
|---|
-Non-polymers , 6 types, 310 molecules 

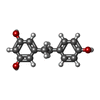








| #2: Chemical | ChemComp-ZN / |
|---|---|
| #3: Chemical | ChemComp-YB / |
| #4: Chemical | ChemComp-RE2 / |
| #5: Chemical | ChemComp-ACT / |
| #6: Chemical | ChemComp-IMD / |
| #7: Water | ChemComp-HOH / |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.41 Å3/Da / Density % sol: 48.93 % |
|---|---|
| Crystal grow | Temperature: 289 K / Method: vapor diffusion, sitting drop / pH: 6.5 Details: 13% PEG 8000, 100 mM Imidazole pH 6.5, 100 mM Na Acetate, 5 mM YbCl3, Soak with 20 mM Dihydroresveratrol, VAPOR DIFFUSION, SITTING DROP, temperature 289K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU MICROMAX-007 HF / Wavelength: 1.54178 Å ROTATING ANODE / Type: RIGAKU MICROMAX-007 HF / Wavelength: 1.54178 Å |
| Detector | Type: RIGAKU SATURN 944 / Detector: CCD / Date: Jun 4, 2007 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.54178 Å / Relative weight: 1 |
| Reflection | Resolution: 1.9→50 Å / Num. obs: 50670 / % possible obs: 94.5 % / Redundancy: 3.8 % / Rmerge(I) obs: 0.063 / Net I/σ(I): 16.8 |
| Reflection shell | Resolution: 1.9→1.97 Å / Redundancy: 2.5 % / Rmerge(I) obs: 0.655 / % possible all: 82.3 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB entry 3FH7 Resolution: 1.9→50 Å / Cor.coef. Fo:Fc: 0.959 / Cor.coef. Fo:Fc free: 0.942 / SU B: 3.4 / SU ML: 0.1 / Cross valid method: THROUGHOUT / ESU R: 0.162 / ESU R Free: 0.148 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 26.46 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.9→50 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.9→1.95 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller

























 PDBj
PDBj










