[English] 日本語
 Yorodumi
Yorodumi- EMDB-36326: Cryo-EM structure of the GLP-1R/GCGR dual agonist peptide15-bound... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the GLP-1R/GCGR dual agonist peptide15-bound human GLP-1R-Gs complex | |||||||||||||||||||||||||||
 Map data Map data | ||||||||||||||||||||||||||||
 Sample Sample |
| |||||||||||||||||||||||||||
 Keywords Keywords | G protein-coupled receptor / ligand recognition / receptor activation / unimolecular dual agonist / STRUCTURAL PROTEIN | |||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationglucagon-like peptide 1 receptor activity / glucagon receptor activity / hormone secretion / positive regulation of blood pressure / G-protein activation / Activation of the phototransduction cascade / Glucagon-type ligand receptors / Thromboxane signalling through TP receptor / Sensory perception of sweet, bitter, and umami (glutamate) taste / G beta:gamma signalling through PI3Kgamma ...glucagon-like peptide 1 receptor activity / glucagon receptor activity / hormone secretion / positive regulation of blood pressure / G-protein activation / Activation of the phototransduction cascade / Glucagon-type ligand receptors / Thromboxane signalling through TP receptor / Sensory perception of sweet, bitter, and umami (glutamate) taste / G beta:gamma signalling through PI3Kgamma / G beta:gamma signalling through CDC42 / Ca2+ pathway / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / post-translational protein targeting to membrane, translocation / G alpha (z) signalling events / Vasopressin regulates renal water homeostasis via Aquaporins / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / Adrenaline,noradrenaline inhibits insulin secretion / ADP signalling through P2Y purinoceptor 12 / G alpha (q) signalling events / Thrombin signalling through proteinase activated receptors (PARs) / G alpha (i) signalling events / Activation of G protein gated Potassium channels / G-protein activation / G beta:gamma signalling through PI3Kgamma / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through PLC beta / ADP signalling through P2Y purinoceptor 1 / Thromboxane signalling through TP receptor / Presynaptic function of Kainate receptors / G beta:gamma signalling through CDC42 / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Glucagon-type ligand receptors / Adrenaline,noradrenaline inhibits insulin secretion / G alpha (12/13) signalling events / G beta:gamma signalling through BTK / ADP signalling through P2Y purinoceptor 12 / alkylglycerophosphoethanolamine phosphodiesterase activity / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / Thrombin signalling through proteinase activated receptors (PARs) / Ca2+ pathway / G alpha (z) signalling events / Extra-nuclear estrogen signaling / G alpha (s) signalling events / G alpha (q) signalling events / response to psychosocial stress / photoreceptor outer segment membrane / G alpha (i) signalling events / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / regulation of heart contraction / spectrin binding / Vasopressin regulates renal water homeostasis via Aquaporins / activation of adenylate cyclase activity / peptide hormone binding / photoreceptor outer segment / cardiac muscle cell apoptotic process / negative regulation of blood pressure / photoreceptor inner segment / cAMP-mediated signaling / adenylate cyclase-activating G protein-coupled receptor signaling pathway / Glucagon-type ligand receptors / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / cellular response to catecholamine stimulus / sensory perception of taste / transmembrane signaling receptor activity / adenylate cyclase-activating dopamine receptor signaling pathway / cellular response to prostaglandin E stimulus / G-protein beta-subunit binding / heterotrimeric G-protein complex / signaling receptor complex adaptor activity / GTPase binding / retina development in camera-type eye / phospholipase C-activating G protein-coupled receptor signaling pathway / positive regulation of cytosolic calcium ion concentration / cellular response to hypoxia / cell body / G alpha (s) signalling events / cell population proliferation / learning or memory / cell surface receptor signaling pathway / G protein-coupled receptor signaling pathway / GTPase activity / dendrite / protein-containing complex binding / membrane / plasma membrane / cytoplasm Similarity search - Function | |||||||||||||||||||||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /    | |||||||||||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.46 Å | |||||||||||||||||||||||||||
 Authors Authors | Yang L / Zhou QT / Dai AT / Zhao FH / Chang RL / Ying TL / Wu BL / Yang DH / Wang MW / Cong ZT | |||||||||||||||||||||||||||
| Funding support |  China, 8 items China, 8 items
| |||||||||||||||||||||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2023 Journal: Proc Natl Acad Sci U S A / Year: 2023Title: Structural analysis of the dual agonism at GLP-1R and GCGR. Authors: Yang Li / Qingtong Zhou / Antao Dai / Fenghui Zhao / Rulue Chang / Tianlei Ying / Beili Wu / Dehua Yang / Ming-Wei Wang / Zhaotong Cong /   Abstract: Glucagon-like peptide-1 receptor (GLP-1R) and glucagon receptor (GCGR), two members of class B1 G protein-coupled receptors, play important roles in glucose homeostasis and energy metabolism. They ...Glucagon-like peptide-1 receptor (GLP-1R) and glucagon receptor (GCGR), two members of class B1 G protein-coupled receptors, play important roles in glucose homeostasis and energy metabolism. They share a high degree of sequence homology but have different functionalities. Unimolecular dual agonists of both receptors developed recently displayed better clinical efficacies than that of monotherapy. To study the underlying molecular mechanisms, we determined high-resolution cryo-electron microscopy structures of GLP-1R or GCGR in complex with heterotrimeric G protein and three GLP-1R/GCGR dual agonists including peptide 15, MEDI0382 (cotadutide) and SAR425899 with variable activating profiles at GLP-1R versus GCGR. Compared with related structures reported previously and supported by our published pharmacological data, key residues responsible for ligand recognition and dual agonism were identified. Analyses of peptide conformational features revealed a difference in side chain orientations within the first three residues, indicating that distinct engagements in the deep binding pocket are required to achieve receptor selectivity. The middle region recognizes extracellular loop 1 (ECL1), ECL2, and the top of transmembrane helix 1 (TM1) resulting in specific conformational changes of both ligand and receptor, especially the dual agonists reshaped ECL1 conformation of GLP-1R relative to that of GCGR, suggesting an important role of ECL1 interaction in executing dual agonism. Structural investigation of lipid modification showed a better interaction between lipid moiety of MEDI0382 and TM1-TM2 cleft, in line with its increased potency at GCGR than SAR425899. Together, the results provide insightful information for the design and development of improved therapeutics targeting these two receptors simultaneously. | |||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_36326.map.gz emd_36326.map.gz | 32.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-36326-v30.xml emd-36326-v30.xml emd-36326.xml emd-36326.xml | 21.3 KB 21.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_36326.png emd_36326.png | 90.7 KB | ||
| Filedesc metadata |  emd-36326.cif.gz emd-36326.cif.gz | 6.6 KB | ||
| Others |  emd_36326_half_map_1.map.gz emd_36326_half_map_1.map.gz emd_36326_half_map_2.map.gz emd_36326_half_map_2.map.gz | 59.4 MB 59.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-36326 http://ftp.pdbj.org/pub/emdb/structures/EMD-36326 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36326 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36326 | HTTPS FTP |
-Validation report
| Summary document |  emd_36326_validation.pdf.gz emd_36326_validation.pdf.gz | 684.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_36326_full_validation.pdf.gz emd_36326_full_validation.pdf.gz | 683.8 KB | Display | |
| Data in XML |  emd_36326_validation.xml.gz emd_36326_validation.xml.gz | 12.3 KB | Display | |
| Data in CIF |  emd_36326_validation.cif.gz emd_36326_validation.cif.gz | 14.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36326 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36326 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36326 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36326 | HTTPS FTP |
-Related structure data
| Related structure data |  8jisMC  8jipC  8jiqC  8jirC  8jitC  8jiuC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_36326.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_36326.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.071 Å | ||||||||||||||||||||||||||||||||||||
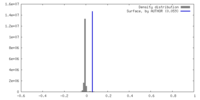
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_36326_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
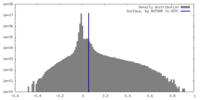
| Density Histograms |
-Half map: #1
| File | emd_36326_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
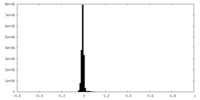
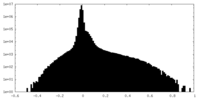
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM structure of the GLP-1R/GCGR dual agonist peptide 15-boun...
| Entire | Name: Cryo-EM structure of the GLP-1R/GCGR dual agonist peptide 15-bound human GLP-1R-Gs complex |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM structure of the GLP-1R/GCGR dual agonist peptide 15-boun...
| Supramolecule | Name: Cryo-EM structure of the GLP-1R/GCGR dual agonist peptide 15-bound human GLP-1R-Gs complex type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short
| Macromolecule | Name: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 41.401863 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: SAEDKAAVER SKMIEKQLQK DKQVYRATHR LLLLGADNSG KSTIVKQMRI YHVNGYSEEE CKQYKAVVYS NTIQSIIAII RAMGRLKID FGDSARADDA RQLFVLAGAA EEGFMTAELA GVIKRLWKDS GVQACFNRSR EYQLNDSAAY YLNDLDRIAQ P NYIPTQQD ...String: SAEDKAAVER SKMIEKQLQK DKQVYRATHR LLLLGADNSG KSTIVKQMRI YHVNGYSEEE CKQYKAVVYS NTIQSIIAII RAMGRLKID FGDSARADDA RQLFVLAGAA EEGFMTAELA GVIKRLWKDS GVQACFNRSR EYQLNDSAAY YLNDLDRIAQ P NYIPTQQD VLRTRVKTSG IFETKFQVDK VNFHMFDVGA QRDERRKWIQ CFNDVTAIIF VVDSSDYNRL QEALNDFKSI WN NRWLRTI SVILFLNKQD LLAEKVLAGK SKIEDYFPEF ARYTTPEDAT PEPGEDPRVT RAKYFIRDEF LRISTASGDG RHY CYPHFT CSVDTENIRR VFNDCRDIIQ RMHLRQYELL |
-Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 37.198656 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: ELDQLRQEAE QLKNQIRDAR KACADATLSQ ITNNIDPVGR IQMRTRRTLR GHLAKIYAMH WGTDSRLLVS ASQDGKLIIW DSYTTNKVH AIPLRSSWVM TCAYAPSGNY VACGGLDNIC SIYNLKTREG NVRVSRELAG HTGYLSCCRF LDDNQIVTSS G DTTCALWD ...String: ELDQLRQEAE QLKNQIRDAR KACADATLSQ ITNNIDPVGR IQMRTRRTLR GHLAKIYAMH WGTDSRLLVS ASQDGKLIIW DSYTTNKVH AIPLRSSWVM TCAYAPSGNY VACGGLDNIC SIYNLKTREG NVRVSRELAG HTGYLSCCRF LDDNQIVTSS G DTTCALWD IETGQQTTTF TGHTGDVMSL SLAPDTRLFV SGACDASAKL WDVREGMCRQ TFTGHESDIN AICFFPNGNA FA TGSDDAT CRLFDLRADQ ELMTYSHDNI ICGITSVSFS KSGRLLLAGY DDFNCNVWDA LKADRAGVLA GHDNRVSCLG VTD DGMAVA TGSWDSFLKI WN UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #3: Peptide 15
| Macromolecule | Name: Peptide 15 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 3.385689 KDa |
| Sequence | String: HSQGTFTSDY SKYLDEQAAK EFIAWLMNT |
-Macromolecule #4: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 6.261229 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: TASIAQARKL VEQLKMEANI DRIKVSKAAA DLMAYCEAHA KEDPLLTPVP ASENPFR UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #5: Nanobody 35
| Macromolecule | Name: Nanobody 35 / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 13.711284 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: QVQLQESGGG LVQPGGSLRL SCAASGFTFS NYKMNWVRQA PGKGLEWVSD ISQSGASISY TGSVKGRFTI SRDNAKNTLY LQMNSLKPE DTAVYYCARC PAPFTRDCFD VTSTTYAYRG QGTQVTV |
-Macromolecule #6: Glucagon-like peptide 1 receptor
| Macromolecule | Name: Glucagon-like peptide 1 receptor / type: protein_or_peptide / ID: 6 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 46.130492 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: VSLWETVQKW REYRRQCQRS LTEDPPPATD LFCNRTFDEY ACWPDGEPGS FVNVSCPWYL PWASSVPQGH VYRFCTAEGL WLQKDNSSL PWRDLSECEE SKRGERSSPE EQLLFLYIIY TVGYALSFSA LVIASAILLG FRHLHCTRNY IHLNLFASFI L RALSVFIK ...String: VSLWETVQKW REYRRQCQRS LTEDPPPATD LFCNRTFDEY ACWPDGEPGS FVNVSCPWYL PWASSVPQGH VYRFCTAEGL WLQKDNSSL PWRDLSECEE SKRGERSSPE EQLLFLYIIY TVGYALSFSA LVIASAILLG FRHLHCTRNY IHLNLFASFI L RALSVFIK DAALKWMYST AAQQHQWDGL LSYQDSLSCR LVFLLMQYCV AANYYWLLVE GVYLYTLLAF SVLSEQWIFR LY VSIGWGV PLLFVVPWGI VKYLYEDEGC WTRNSNMNYW LIIRLPILFA IGVNFLIFVR VICIVVSKLK ANLMCKTDIK CRL AKSTLT LIPLLGTHEV IFAFVMDEHA RGTLRFIKLF TELSFTSFQG LMVAILYCFV NNEVQLEFRK SWERWRLE UniProtKB: Glucagon-like peptide 1 receptor |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 80.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source: OTHER |
| Electron optics | Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.2 µm / Nominal defocus min: 1.2 µm |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.46 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 1252175 |
| Initial angle assignment | Type: OTHER |
| Final angle assignment | Type: OTHER |
 Movie
Movie Controller
Controller





















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)