[English] 日本語
 Yorodumi
Yorodumi- EMDB-35084: Composite cryo-EM structure of the histone deacetylase complex Rp... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
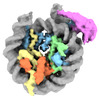
| Title | Composite cryo-EM structure of the histone deacetylase complex Rpd3S in complex with nucleosome | |||||||||
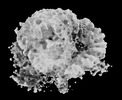
 Map data Map data | Composite EM map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Histone deacetylase complex / nucleosome / GENE REGULATION | |||||||||
| Function / homology |  Function and homology information Function and homology informationSnt2C complex / negative regulation of reciprocal meiotic recombination / negative regulation of silent mating-type cassette heterochromatin formation / Rpd3L complex / protein localization to nucleolar rDNA repeats / Rpd3L-Expanded complex / negative regulation of rDNA heterochromatin formation / Rpd3S complex / rDNA chromatin condensation / nucleophagy ...Snt2C complex / negative regulation of reciprocal meiotic recombination / negative regulation of silent mating-type cassette heterochromatin formation / Rpd3L complex / protein localization to nucleolar rDNA repeats / Rpd3L-Expanded complex / negative regulation of rDNA heterochromatin formation / Rpd3S complex / rDNA chromatin condensation / nucleophagy / HDACs deacetylate histones / histone deacetylase / cellular response to nitrogen starvation / SUMOylation of chromatin organization proteins / regulation of DNA-templated DNA replication initiation / negative regulation of transcription by RNA polymerase I / histone deacetylase activity / NuA4 histone acetyltransferase complex / Sin3-type complex / Estrogen-dependent gene expression / histone deacetylase complex / positive regulation of macroautophagy / meiotic cell cycle / nuclear periphery / transcription elongation by RNA polymerase II / G1/S transition of mitotic cell cycle / heterochromatin formation / G2/M transition of mitotic cell cycle / double-strand break repair via nonhomologous end joining / structural constituent of chromatin / transcription corepressor activity / nucleosome / nucleosome assembly / cellular response to heat / transcription coactivator activity / response to oxidative stress / chromatin remodeling / protein heterodimerization activity / cell division / DNA repair / regulation of transcription by RNA polymerase II / chromatin / regulation of DNA-templated transcription / negative regulation of transcription by RNA polymerase II / positive regulation of transcription by RNA polymerase II / DNA binding / identical protein binding / nucleus / metal ion binding / cytoplasm Similarity search - Function | |||||||||
| Biological species |  | |||||||||
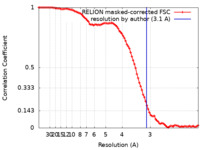
| Method | single particle reconstruction / cryo EM / Resolution: 3.1 Å | |||||||||
 Authors Authors | Cui H / Wang H | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2023 Journal: Nat Struct Mol Biol / Year: 2023Title: Structure of histone deacetylase complex Rpd3S bound to nucleosome. Authors: Wulong Li / Hengjun Cui / Zhimin Lu / Haibo Wang /  Abstract: Crosstalk between histone modifications represents a fundamental epigenetic mechanism in gene regulation. During the transcription elongation process, the histone deacetylase complex Rpd3S is ...Crosstalk between histone modifications represents a fundamental epigenetic mechanism in gene regulation. During the transcription elongation process, the histone deacetylase complex Rpd3S is recruited to H3K36-methylated nucleosomes to suppress cryptic transcription initiation. However, how subunits of Rpd3S are assembled and coordinated to recognize nucleosomal substrates and exert their deacetylation function remains unclear. Here we report the structure of Saccharomyces cerevisiae Rpd3S deacetylase bound to H3K36me3-modified nucleosome at 3.1 Å resolution. It shows that Sin3 and Rco1 subunits orchestrate the assembly of the complex and mediate its contact with nucleosome at multiple sites, with the Sin3-DNA interface as a pivotal anchor. The PHD1 domain of Rco1 recognizes the unmodified H3K4 and places the following H3 tail toward the active site of Rpd3, while the chromodomain of Eaf3 subunit recognizes the H3K36me3 mark and contacts both nucleosomal and linker DNA. The second copy of Eaf3-Rco1 is involved in neighboring nucleosome binding. Our work unravels the structural basis of chromatin targeting and deacetylation by the Rpd3S complex. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_35084.map.gz emd_35084.map.gz | 47 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-35084-v30.xml emd-35084-v30.xml emd-35084.xml emd-35084.xml | 31.1 KB 31.1 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_35084_fsc.xml emd_35084_fsc.xml | 9.1 KB | Display |  FSC data file FSC data file |
| Images |  emd_35084.png emd_35084.png | 177.2 KB | ||
| Filedesc metadata |  emd-35084.cif.gz emd-35084.cif.gz | 9.3 KB | ||
| Others |  emd_35084_half_map_1.map.gz emd_35084_half_map_1.map.gz emd_35084_half_map_2.map.gz emd_35084_half_map_2.map.gz | 59.1 MB 59 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-35084 http://ftp.pdbj.org/pub/emdb/structures/EMD-35084 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35084 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35084 | HTTPS FTP |
-Related structure data
| Related structure data |  8hy0MC  8hxxC  8hxyC  8hxzC  8jhoC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_35084.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_35084.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Composite EM map | ||||||||||||||||||||
| Voxel size | X=Y=Z: 1.05 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Composite EM half2 map
| File | emd_35084_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Composite EM half2 map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Composite EM half2 map
| File | emd_35084_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Composite EM half2 map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Rpd3S histone deacetylase in complex with nucleosome
+Supramolecule #1: Rpd3S histone deacetylase in complex with nucleosome
+Macromolecule #1: Histone H3
+Macromolecule #2: Histone H4
+Macromolecule #3: Histone H2A
+Macromolecule #4: Histone H2B
+Macromolecule #7: Transcriptional regulatory protein SIN3
+Macromolecule #8: Histone deacetylase RPD3
+Macromolecule #9: Chromatin modification-related protein EAF3
+Macromolecule #10: RCO1 isoform 1
+Macromolecule #5: DNA (352-MER)
+Macromolecule #6: DNA (352-MER)
+Macromolecule #11: ZINC ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 Details: 20 mM HEPES-Na pH 7.5, 40 mM KCl, 2 mM MgCl2, 1 mM TCEP |
|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 44.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.8 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
|---|---|
| Output model |  PDB-8hy0: |
 Movie
Movie Controller
Controller















 Z
Z Y
Y X
X


















 Trichoplusia ni (cabbage looper)
Trichoplusia ni (cabbage looper)