+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | DmDcr-2/R2D2/LoqsPD with 19bp-dsRNA in Trimer state | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Ribonuclease / DOUBLE STRANDED RNA / RNA BINDING PROTEIN-RNA COMPLEX | |||||||||
| Function / homology |  Function and homology information Function and homology informationfollicle cell of egg chamber stalk formation / lncRNA catabolic process / : / positive regulation of Toll signaling pathway / MicroRNA (miRNA) biogenesis / RNAi-mediated antiviral immune response / Small interfering RNA (siRNA) biogenesis / female germ-line stem cell asymmetric division / PKR-mediated signaling / regulation of regulatory ncRNA processing ...follicle cell of egg chamber stalk formation / lncRNA catabolic process / : / positive regulation of Toll signaling pathway / MicroRNA (miRNA) biogenesis / RNAi-mediated antiviral immune response / Small interfering RNA (siRNA) biogenesis / female germ-line stem cell asymmetric division / PKR-mediated signaling / regulation of regulatory ncRNA processing / dsRNA transport / dosage compensation by hyperactivation of X chromosome / RISC complex binding / global gene silencing by mRNA cleavage / germ-line stem cell population maintenance / apoptotic DNA fragmentation / ribonuclease III / deoxyribonuclease I activity / RISC-loading complex / miRNA metabolic process / detection of virus / RISC complex assembly / regulatory ncRNA-mediated post-transcriptional gene silencing / ribonuclease III activity / pre-miRNA processing / siRNA binding / siRNA processing / ATP-dependent activity, acting on RNA / positive regulation of innate immune response / RISC complex / positive regulation of defense response to virus by host / helicase activity / central nervous system development / mRNA 3'-UTR binding / locomotory behavior / heterochromatin formation / cellular response to virus / cytoplasmic ribonucleoprotein granule / double-stranded RNA binding / defense response to virus / perinuclear region of cytoplasm / ATP hydrolysis activity / RNA binding / ATP binding / nucleus / cytosol / cytoplasm Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.7 Å | |||||||||
 Authors Authors | Su S / Wang J / Ma J | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2023 Journal: Nat Commun / Year: 2023Title: Structural mechanism of R2D2 and Loqs-PD synergistic modulation on DmDcr-2 oligomers. Authors: Ting Deng / Shichen Su / Xun Yuan / Jinqiu He / Ying Huang / Jinbiao Ma / Jia Wang /  Abstract: Small interference RNAs are the key components of RNA interference, a conserved RNA silencing or viral defense mechanism in many eukaryotes. In Drosophila melanogaster, Dicer-2 (DmDcr-2)-mediated ...Small interference RNAs are the key components of RNA interference, a conserved RNA silencing or viral defense mechanism in many eukaryotes. In Drosophila melanogaster, Dicer-2 (DmDcr-2)-mediated RNAi pathway plays important roles in defending against viral infections and protecting genome integrity. During the maturation of siRNAs, two cofactors can regulate DmDcr-2's functions: Loqs-PD that is required for dsRNA processing, and R2D2 that is essential for the subsequent loading of siRNAs into effector Ago2 to form RISC complexes. However, due to the lack of structural information, it is still unclear whether R2D2 and Loqs-PD affect the functions of DmDcr-2 simultaneously. Here we present several cryo-EM structures of DmDcr-2/R2D2/Loqs-PD complex bound to dsRNAs with various lengths by the Helicase domain. These structures revealed that R2D2 and Loqs-PD can bind to different regions of DmDcr-2 without interfering with each other. Furthermore, the cryo-EM results demonstrate that these complexes can form large oligomers and assemble into fibers. The formation and depolymerization of these oligomers are associated with ATP hydrolysis. These findings provide insights into the structural mechanism of DmDcr-2 and its cofactors during siRNA processing. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_34708.map.gz emd_34708.map.gz | 118 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-34708-v30.xml emd-34708-v30.xml emd-34708.xml emd-34708.xml | 19.2 KB 19.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_34708.png emd_34708.png | 98.7 KB | ||
| Others |  emd_34708_half_map_1.map.gz emd_34708_half_map_1.map.gz emd_34708_half_map_2.map.gz emd_34708_half_map_2.map.gz | 116 MB 116 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-34708 http://ftp.pdbj.org/pub/emdb/structures/EMD-34708 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34708 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34708 | HTTPS FTP |
-Validation report
| Summary document |  emd_34708_validation.pdf.gz emd_34708_validation.pdf.gz | 1.2 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_34708_full_validation.pdf.gz emd_34708_full_validation.pdf.gz | 1.2 MB | Display | |
| Data in XML |  emd_34708_validation.xml.gz emd_34708_validation.xml.gz | 14.1 KB | Display | |
| Data in CIF |  emd_34708_validation.cif.gz emd_34708_validation.cif.gz | 16.5 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34708 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34708 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34708 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34708 | HTTPS FTP |
-Related structure data
| Related structure data |  8hf1MC  8hf0C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_34708.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_34708.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.08 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_34708_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
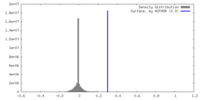
| Density Histograms |
-Half map: #1
| File | emd_34708_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
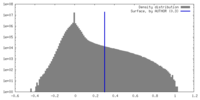
| Density Histograms |
- Sample components
Sample components
-Entire : DmDcr-2/R2D2/LoqsPD with 19bp-dsRNA in Trimer state
| Entire | Name: DmDcr-2/R2D2/LoqsPD with 19bp-dsRNA in Trimer state |
|---|---|
| Components |
|
-Supramolecule #1: DmDcr-2/R2D2/LoqsPD with 19bp-dsRNA in Trimer state
| Supramolecule | Name: DmDcr-2/R2D2/LoqsPD with 19bp-dsRNA in Trimer state / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 300 KDa |
-Macromolecule #1: Dicer-2, isoform A
| Macromolecule | Name: Dicer-2, isoform A / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO / EC number: deoxyribonuclease I |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 197.875484 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: EDVEIKPRGY QLRLVDHLTK SNGIVYLPTG SGKTFVAILV LKRFSQDFDK PIESGGKRAL FMCNTVELAR QQAMAVRRCT NFKVGFYVG EQGVDDWTRG MWSDEIKKNQ VLVGTAQVFL DMVTQTYVAL SSLSVVIIDE CHHGTGHHPF REFMRLFTIA N QTKLPRVV ...String: EDVEIKPRGY QLRLVDHLTK SNGIVYLPTG SGKTFVAILV LKRFSQDFDK PIESGGKRAL FMCNTVELAR QQAMAVRRCT NFKVGFYVG EQGVDDWTRG MWSDEIKKNQ VLVGTAQVFL DMVTQTYVAL SSLSVVIIDE CHHGTGHHPF REFMRLFTIA N QTKLPRVV GLTGVLIKGN EITNVATKLK ELEITYRGNI ITVSDTKEME NVMLYATKPT EVMVSFPHQE QVLTVTRLIS AE IEKFYVS LDLMNIGVQP IRRSKSLQCL RDPSKKSFVK QLFNDFLYQM KEYGIYAASI AIISLIVEFD IKRRQAETLS VKL MHRTAL TLCEKIRHLL VQKLQDMTYD DDDDNVNTEE VIMNFSTPKV QRFLMSLKVS FADKDPKDIC CLVFVERRYT CKCI YGLLL NYIQSTPELR NVLTPQFMVG RNNISPDFES VLERKWQKSA IQQFRDGNAN LMICSSVLEE GIDVQACNHV FILDP VKTF NMYVQSKGRA RTTEAKFVLF TADKEREKTI QQIYQYRKAH NDIAEYLKDR VLEKTEPELY EIKGHFQDDI DPFTNE NGA VLLPNNALAI LHRYCQTIPT DAFGFVIPWF HVLQEDERDR IFGVSAKGKH VISINMPVNC MLRDTIYSDP MDNVKTA KI SAAFKACKVL YSLGELNERF VPKTLKERVA SIADVHFEHW NKYGDSVTAT VNKADKSKDR TYKTECPLEF YDALPRVG E ICYAYEIFLE PQFESCEYTE HMYLNLQTPR NYAILLRNKL PRLAEMPLFS NQGKLHVRVA NAPLEVIIQN SEQLELLHQ FHGMVFRDIL KIWHPFFVLD RRSKENSYLV VPLILGAGEQ KCFDWELMTN FRRLPQSHGS NVQQREQQPA PRPEDFEGKI VTQWYANYD KPMLVTKVHR ELTPLSYMEK NQQDKTYYEF TMSKYGNRIG DVVHKDKFMI EVRDLTEQLT FYVHNRGKFN A KSKAKMKV ILIPELCFNF NFPGDLWLKL IFLPSILNRM YFLLHAEALR KRFNTYLNLH LLPFNGTDYM PRPLEIDYSL KR NVDPLGN VIPTEDIEEP KSLLEPMPTK SIEASVANLE ITEFENPWQK YMEPVDLSRN LLSTYPVELD YYYHFSVGNV CEM NEMDFE DKEYWAKNQF HMPTGNIYGN RTPAKTNANV PALMPSKPTV RGKVKPLLIL QKTVSKEHIT PAEQGEFLAA ITAS SAADV FDMERLEILG NSFLKLSATL YLASKYSDWN EGTLTEVKSK LVSNRNLLFC LIDADIPKTL NTIQFTPRYT WLPPG ISLP HNVLALWREN PEFAKIIGPH NLRDLALGDE ESLVKGNCSD INYNRFVEGC RANGQSFYAG ADFSSEVNFC VGLVTI PNK VIADTLEALL GVIVKNYGLQ HAFKMLEYFK ICRADIDKPL TQLLNLELGG KKMRANVNTT EIDGFLINHY YLEKNLG YT FKDRRYLLQA LTHPSYPTNR ITGSYQELEF IGNAILDFLI SAYIFENNTK MNPGALTDLR SALVNNTTLA CICVRHRL H FFILAENAKL SEIISKFVNF QESQGHRVTN YVRILLEEAD VQPTPLDLDD ELDMTELPHA NKCISQEAEK GVPPKGEFN MSTNVDVPKA LGDVLEALIA AVYLDCRDLQ RTWEVIFNLF EPELQEFTRK VPINHIRQLV EHKHAKPVFS SPIVEGETVM VSCQFTCME KTIKVYGFGS NKDQAKLSAA KHALQQLSKC DA UniProtKB: Endoribonuclease Dcr-2 |
-Macromolecule #2: Loquacious, isoform D
| Macromolecule | Name: Loquacious, isoform D / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 2.033304 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: IDRYEQVSKD FEFIKI UniProtKB: Protein Loquacious |
-Macromolecule #3: LD06392p
| Macromolecule | Name: LD06392p / type: protein_or_peptide / ID: 3 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 14.706812 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MEESMEELEA LRRKKFTTYW ELKEAGSVDH TGMRLCDRHN YFKNFYPTLK KEAIEAINSD EYESSKDKAM DVMSSLKITP KISEVESSS LVPLLSVELN CAFDVVLMAK ETDIYDHIID YFRTMLI UniProtKB: LD06392p |
-Macromolecule #4: RNA (5'-R(P*GP*AP*GP*AP*CP*UP*UP*GP*G)-3')
| Macromolecule | Name: RNA (5'-R(P*GP*AP*GP*AP*CP*UP*UP*GP*G)-3') / type: rna / ID: 4 / Number of copies: 3 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 2.911791 KDa |
| Sequence | String: GAGACUUGG |
-Macromolecule #5: RNA (5'-R(P*CP*CP*AP*AP*GP*UP*CP*UP*CP*UP*U)-3')
| Macromolecule | Name: RNA (5'-R(P*CP*CP*AP*AP*GP*UP*CP*UP*CP*UP*U)-3') / type: rna / ID: 5 / Number of copies: 3 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 3.40405 KDa |
| Sequence | String: CCAAGUCUCU U |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.3 mg/mL |
|---|---|
| Buffer | pH: 8 |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 0.01 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Algorithm: FOURIER SPACE / Resolution.type: BY AUTHOR / Resolution: 3.7 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: cryoSPARC (ver. 3.2) / Number images used: 46899 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
-Atomic model buiding 1
| Refinement | Protocol: RIGID BODY FIT |
|---|---|
| Output model |  PDB-8hf1: |
 Movie
Movie Controller
Controller








 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)