Entry Database : PDB / ID : 2x1eTitle The crystal structure of mature acyl coenzyme A:isopenicillin N acyltransferase from Penicillium chrysogenum in complex 6- aminopenicillanic acid ACYL-COENZYME Keywords / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / Biological species PENICILLIUM CHRYSOGENUM (fungus)Method / / / Resolution : 2 Å Authors Bokhove, M. / Yoshida, H. / Hensgens, C.M.H. / van der Laan, J.M. / Sutherland, J.D. / Dijkstra, B.W. Journal : Structure / Year : 2010Title : Structures of an Isopenicillin N Converting Ntn-Hydrolase Reveal Different Catalytic Roles for the Active Site Residues of Precursor and Mature Enzyme.Authors : Bokhove, M. / Yoshida, H. / Hensgens, C.M.H. / Van Der Laan, J.M. / Sutherland, J.D. / Dijkstra, B.W. History Deposition Dec 23, 2009 Deposition site / Processing site Revision 1.0 Mar 9, 2010 Provider / Type Revision 1.1 Dec 28, 2011 Group Database references / Derived calculations ... Database references / Derived calculations / Non-polymer description / Refinement description / Version format compliance Revision 1.2 Nov 5, 2014 Group Atomic model / Derived calculations ... Atomic model / Derived calculations / Non-polymer description / Other / Structure summary Revision 1.3 Jul 24, 2019 Group / Derived calculations / Category / struct_connItem / _struct_conn.pdbx_leaving_atom_flagRevision 1.4 Dec 20, 2023 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Other / Refinement description Category chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_database_status / pdbx_initial_refinement_model / struct_site Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_database_status.status_code_sf / _struct_site.pdbx_auth_asym_id / _struct_site.pdbx_auth_comp_id / _struct_site.pdbx_auth_seq_id Revision 1.5 Nov 13, 2024 Group / Category / pdbx_modification_feature
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information PENICILLIUM CHRYSOGENUM (fungus)
PENICILLIUM CHRYSOGENUM (fungus) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2 Å
MOLECULAR REPLACEMENT / Resolution: 2 Å  Authors
Authors Citation
Citation Journal: Structure / Year: 2010
Journal: Structure / Year: 2010 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 2x1e.cif.gz
2x1e.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb2x1e.ent.gz
pdb2x1e.ent.gz PDB format
PDB format 2x1e.json.gz
2x1e.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/x1/2x1e
https://data.pdbj.org/pub/pdb/validation_reports/x1/2x1e ftp://data.pdbj.org/pub/pdb/validation_reports/x1/2x1e
ftp://data.pdbj.org/pub/pdb/validation_reports/x1/2x1e
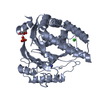
 Links
Links Assembly
Assembly
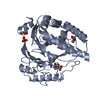



 Components
Components PENICILLIUM CHRYSOGENUM (fungus) / Strain: AS-P-78 / Plasmid: PKC787 / Production host:
PENICILLIUM CHRYSOGENUM (fungus) / Strain: AS-P-78 / Plasmid: PKC787 / Production host: 








 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  EMBL/DESY, HAMBURG
EMBL/DESY, HAMBURG  / Beamline: BW7A / Wavelength: 0.976
/ Beamline: BW7A / Wavelength: 0.976  Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller












 PDBj
PDBj


