+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human Oct4 bound to nucleosome with human nMatn1 sequence | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Oct4 / Nucleosome / Histone / nMatn1 DNA / DNA BINDING PROTEIN-DNA complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationcell fate commitment involved in formation of primary germ layer / cardiac cell fate determination / POU5F1 (OCT4), SOX2, NANOG repress genes related to differentiation / Formation of the anterior neural plate / endodermal-mesodermal cell signaling / endodermal cell fate specification / regulation of asymmetric cell division / Specification of primordial germ cells / heart induction / POU5F1 (OCT4), SOX2, NANOG activate genes related to proliferation ...cell fate commitment involved in formation of primary germ layer / cardiac cell fate determination / POU5F1 (OCT4), SOX2, NANOG repress genes related to differentiation / Formation of the anterior neural plate / endodermal-mesodermal cell signaling / endodermal cell fate specification / regulation of asymmetric cell division / Specification of primordial germ cells / heart induction / POU5F1 (OCT4), SOX2, NANOG activate genes related to proliferation / Specification of the neural plate border / Transcriptional regulation of pluripotent stem cells / Germ layer formation at gastrulation / miRNA binding / somatic stem cell population maintenance / blastocyst development / anatomical structure morphogenesis / BMP signaling pathway / negative regulation of miRNA transcription / DNA-binding transcription repressor activity, RNA polymerase II-specific / response to wounding / structural constituent of chromatin / sequence-specific double-stranded DNA binding / nucleosome / positive regulation of canonical Wnt signaling pathway / nucleosome assembly / regulation of gene expression / RNA polymerase II-specific DNA-binding transcription factor binding / transcription regulator complex / sequence-specific DNA binding / transcription cis-regulatory region binding / DNA-binding transcription factor activity, RNA polymerase II-specific / protein heterodimerization activity / DNA-binding transcription factor activity / RNA polymerase II cis-regulatory region sequence-specific DNA binding / ubiquitin protein ligase binding / regulation of DNA-templated transcription / regulation of transcription by RNA polymerase II / negative regulation of transcription by RNA polymerase II / positive regulation of transcription by RNA polymerase II / mitochondrion / DNA binding / RNA binding / nucleoplasm / nucleus / cytosol / cytoplasm Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.3 Å | |||||||||
 Authors Authors | Sinha KK / Bilokapic S / Du Y / Malik D / Halic M | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
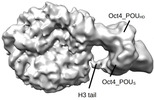
 Citation Citation |  Journal: Nature / Year: 2023 Journal: Nature / Year: 2023Title: Histone modifications regulate pioneer transcription factor cooperativity. Authors: Kalyan K Sinha / Silvija Bilokapic / Yongming Du / Deepshikha Malik / Mario Halic /  Abstract: Pioneer transcription factors have the ability to access DNA in compacted chromatin. Multiple transcription factors can bind together to a regulatory element in a cooperative way, and cooperation ...Pioneer transcription factors have the ability to access DNA in compacted chromatin. Multiple transcription factors can bind together to a regulatory element in a cooperative way, and cooperation between the pioneer transcription factors OCT4 (also known as POU5F1) and SOX2 is important for pluripotency and reprogramming. However, the molecular mechanisms by which pioneer transcription factors function and cooperate on chromatin remain unclear. Here we present cryo-electron microscopy structures of human OCT4 bound to a nucleosome containing human LIN28B or nMATN1 DNA sequences, both of which bear multiple binding sites for OCT4. Our structural and biochemistry data reveal that binding of OCT4 induces changes to the nucleosome structure, repositions the nucleosomal DNA and facilitates cooperative binding of additional OCT4 and of SOX2 to their internal binding sites. The flexible activation domain of OCT4 contacts the N-terminal tail of histone H4, altering its conformation and thus promoting chromatin decompaction. Moreover, the DNA-binding domain of OCT4 engages with the N-terminal tail of histone H3, and post-translational modifications at H3K27 modulate DNA positioning and affect transcription factor cooperativity. Thus, our findings suggest that the epigenetic landscape could regulate OCT4 activity to ensure proper cell programming. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_29843.map.gz emd_29843.map.gz | 23.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-29843-v30.xml emd-29843-v30.xml emd-29843.xml emd-29843.xml | 24.3 KB 24.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_29843.png emd_29843.png | 74.4 KB | ||
| Filedesc metadata |  emd-29843.cif.gz emd-29843.cif.gz | 6.8 KB | ||
| Others |  emd_29843_half_map_1.map.gz emd_29843_half_map_1.map.gz emd_29843_half_map_2.map.gz emd_29843_half_map_2.map.gz | 23.5 MB 23.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-29843 http://ftp.pdbj.org/pub/emdb/structures/EMD-29843 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29843 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29843 | HTTPS FTP |
-Validation report
| Summary document |  emd_29843_validation.pdf.gz emd_29843_validation.pdf.gz | 923.7 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_29843_full_validation.pdf.gz emd_29843_full_validation.pdf.gz | 923.2 KB | Display | |
| Data in XML |  emd_29843_validation.xml.gz emd_29843_validation.xml.gz | 10.6 KB | Display | |
| Data in CIF |  emd_29843_validation.cif.gz emd_29843_validation.cif.gz | 12.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29843 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29843 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29843 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29843 | HTTPS FTP |
-Related structure data
| Related structure data |  8g88MC  8g86C  8g87C  8g8bC  8g8eC  8g8gC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_29843.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_29843.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.61 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_29843_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_29843_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Nucleosome with Human nMatn1 sequence
| Entire | Name: Nucleosome with Human nMatn1 sequence |
|---|---|
| Components |
|
-Supramolecule #1: Nucleosome with Human nMatn1 sequence
| Supramolecule | Name: Nucleosome with Human nMatn1 sequence / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#7 |
|---|---|
| Source (natural) | Organism: |
-Macromolecule #1: Histone H3
| Macromolecule | Name: Histone H3 / type: protein_or_peptide / ID: 1 / Details: Histone H3.2|Xenopus laevis / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 15.30393 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: ARTKQTARKS TGGKAPRKQL ATKAARKSAP ATGGVKKPHR YRPGTVALRE IRRYQKSTEL LIRKLPFQRL VREIAQDFKT DLRFQSSAV MALQEASEAY LVALFEDTNL CAIHAKRVTI MPKDIQLARR IRGERA UniProtKB: Histone H3.2 |
-Macromolecule #2: Histone H4
| Macromolecule | Name: Histone H4 / type: protein_or_peptide / ID: 2 / Details: Histone H4|Xenopus laevis / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 11.263231 KDa |
| Recombinant expression | Organism:  Escherichia phage HY01 (virus) Escherichia phage HY01 (virus) |
| Sequence | String: SGRGKGGKGL GKGGAKRHRK VLRDNIQGIT KPAIRRLARR GGVKRISGLI YEETRGVLKV FLENVIRDAV TYTEHAKRKT VTAMDVVYA LKRQGRTLYG FGG UniProtKB: Histone H4 |
-Macromolecule #3: Histone H2A
| Macromolecule | Name: Histone H2A / type: protein_or_peptide / ID: 3 / Details: Histone H2A|Xenopus laevis / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 13.978241 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: SGRGKQGGKT RAKAKTRSSR AGLQFPVGRV HRLLRKGNYA ERVGAGAPVY LAAVLEYLTA EILELAGNAA RDNKKTRIIP RHLQLAVRN DEELNKLLGR VTIAQGGVLP NIQSVLLPKK TESSKSAKSK UniProtKB: Histone H2A |
-Macromolecule #4: Histone H2B
| Macromolecule | Name: Histone H2B / type: protein_or_peptide / ID: 4 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 13.524752 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: AKSAPAPKKG SKKAVTKTQK KDGKKRRKTR KESYAIYVYK VLKQVHPDTG ISSKAMSIMN SFVNDVFERI AGEASRLAHY NKRSTITSR EIQTAVRLLL PGELAKHAVS EGTKAVTKYT SAK UniProtKB: Histone H2B 1.1 |
-Macromolecule #7: POU domain, class 5, transcription factor 1
| Macromolecule | Name: POU domain, class 5, transcription factor 1 / type: protein_or_peptide / ID: 7 / Details: human Oct4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 42.299453 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GSSHHHHHHS SGLVPRGSHM ASMTGGQQMG RDPNSMAGHL ASDFAFSPPP GGGGDGPGGP EPGWVDPRTW LSFQGPPGGP GIGPGVGPG SEVWGIPPCP PPYEFCGGMA YCGPQVGVGL VPQGGLETSQ PEGEAGVGVE SNSDGASPEP CTVTPGAVKL E KEKLEQNP ...String: GSSHHHHHHS SGLVPRGSHM ASMTGGQQMG RDPNSMAGHL ASDFAFSPPP GGGGDGPGGP EPGWVDPRTW LSFQGPPGGP GIGPGVGPG SEVWGIPPCP PPYEFCGGMA YCGPQVGVGL VPQGGLETSQ PEGEAGVGVE SNSDGASPEP CTVTPGAVKL E KEKLEQNP EESQDIKALQ KELEQFAKLL KQKRITLGYT QADVGLTLGV LFGKVFSQTT ICRFEALQLS FKNMCKLRPL LQ KWVEEAD NNENLQEICK AETLVQARKR KRTSIENRVR GNLENLFLQC PKPTLQQISH IAQQLGLEKD VVRVWFCNRR QKG KRSSSD YAQREDFEAA GSPFSGGPVS FPLAPGPHFG TPGYGSPHFT ALYSSVPFPE GEAFPPVSVT TLGSPMHSN UniProtKB: POU domain, class 5, transcription factor 1 |
-Macromolecule #5: nMATn1 DNA top strand (168-MER)
| Macromolecule | Name: nMATn1 DNA top strand (168-MER) / type: dna / ID: 5 / Details: nMATn1 DNA top strand / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 56.93359 KDa |
| Sequence | String: (DA)(DC)(DA)(DT)(DG)(DC)(DA)(DC)(DA)(DC) (DA)(DT)(DG)(DC)(DT)(DA)(DA)(DT)(DA)(DT) (DA)(DT)(DG)(DC)(DA)(DC)(DA)(DC)(DA) (DA)(DT)(DG)(DC)(DA)(DC)(DA)(DC)(DA)(DG) (DG) (DT)(DT)(DA)(DA)(DT)(DA) ...String: (DA)(DC)(DA)(DT)(DG)(DC)(DA)(DC)(DA)(DC) (DA)(DT)(DG)(DC)(DT)(DA)(DA)(DT)(DA)(DT) (DA)(DT)(DG)(DC)(DA)(DC)(DA)(DC)(DA) (DA)(DT)(DG)(DC)(DA)(DC)(DA)(DC)(DA)(DG) (DG) (DT)(DT)(DA)(DA)(DT)(DA)(DT)(DA) (DT)(DA)(DC)(DA)(DC)(DA)(DT)(DA)(DC)(DA) (DC)(DA) (DC)(DA)(DC)(DA)(DT)(DG)(DC) (DA)(DC)(DA)(DC)(DA)(DC)(DA)(DC)(DG)(DT) (DG)(DC)(DA) (DC)(DA)(DC)(DA)(DT)(DA) (DT)(DA)(DT)(DG)(DC)(DA)(DC)(DA)(DT)(DG) (DC)(DA)(DT)(DG) (DC)(DA)(DC)(DA)(DC) (DA)(DC)(DG)(DT)(DA)(DT)(DA)(DT)(DG)(DC) (DA)(DC)(DA)(DC)(DA) (DC)(DA)(DT)(DG) (DC)(DA)(DC)(DA)(DT)(DG)(DC)(DA)(DT)(DG) (DC)(DG)(DC)(DA)(DC)(DA) (DT)(DA)(DG) (DT)(DC)(DA)(DC)(DA)(DC)(DA)(DC)(DA)(DT) (DG)(DC)(DA)(DC)(DA)(DC)(DA) (DT)(DT) (DA)(DG)(DC)(DA)(DT)(DA)(DT)(DG)(DC)(DA) (DT)(DA)(DC)(DA)(DC)(DA)(DT)(DA) (DC) (DA)(DT)(DG)(DC)(DA) |
-Macromolecule #6: nMatn1 DNA bottom strand (168-MER)
| Macromolecule | Name: nMatn1 DNA bottom strand (168-MER) / type: dna / ID: 6 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 57.889855 KDa |
| Sequence | String: (DT)(DG)(DC)(DA)(DT)(DG)(DT)(DA)(DT)(DG) (DT)(DG)(DT)(DA)(DT)(DG)(DC)(DA)(DT)(DA) (DT)(DG)(DC)(DT)(DA)(DA)(DT)(DG)(DT) (DG)(DT)(DG)(DC)(DA)(DT)(DG)(DT)(DG)(DT) (DG) (DT)(DG)(DA)(DC)(DT)(DA) ...String: (DT)(DG)(DC)(DA)(DT)(DG)(DT)(DA)(DT)(DG) (DT)(DG)(DT)(DA)(DT)(DG)(DC)(DA)(DT)(DA) (DT)(DG)(DC)(DT)(DA)(DA)(DT)(DG)(DT) (DG)(DT)(DG)(DC)(DA)(DT)(DG)(DT)(DG)(DT) (DG) (DT)(DG)(DA)(DC)(DT)(DA)(DT)(DG) (DT)(DG)(DC)(DG)(DC)(DA)(DT)(DG)(DC)(DA) (DT)(DG) (DT)(DG)(DC)(DA)(DT)(DG)(DT) (DG)(DT)(DG)(DT)(DG)(DC)(DA)(DT)(DA)(DT) (DA)(DC)(DG) (DT)(DG)(DT)(DG)(DT)(DG) (DC)(DA)(DT)(DG)(DC)(DA)(DT)(DG)(DT)(DG) (DC)(DA)(DT)(DA) (DT)(DA)(DT)(DG)(DT) (DG)(DT)(DG)(DC)(DA)(DC)(DG)(DT)(DG)(DT) (DG)(DT)(DG)(DT)(DG) (DC)(DA)(DT)(DG) (DT)(DG)(DT)(DG)(DT)(DG)(DT)(DA)(DT)(DG) (DT)(DG)(DT)(DA)(DT)(DA) (DT)(DA)(DT) (DT)(DA)(DA)(DC)(DC)(DT)(DG)(DT)(DG)(DT) (DG)(DC)(DA)(DT)(DT)(DG)(DT) (DG)(DT) (DG)(DC)(DA)(DT)(DA)(DT)(DA)(DT)(DT)(DA) (DG)(DC)(DA)(DT)(DG)(DT)(DG)(DT) (DG) (DC)(DA)(DT)(DG)(DT) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.2 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
Details: 50 mM HEPES pH 7.5, 1 mM DTT | |||||||||
| Grid | Model: Quantifoil / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY | |||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 95 % / Chamber temperature: 283 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.7000000000000001 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: EMDB MAP EMDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.3 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 4000 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller
















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)