登録情報 データベース : EMDB / ID : EMD-29825タイトル Cryo-EM structure of RNP inter Cryo-EM structure of RNP inter 複合体 : Complex of RNP interタンパク質・ペプチド : Antiviral innate immune response receptor RIG-Iタンパク質・ペプチド : E3 ubiquitin-protein ligase RNF135RNA : p3dsRNA24aRNA : p3dsRNA24bリガンド : ZINC ION / / / / / / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
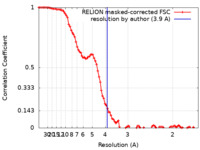
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Homo sapiens (ヒト)手法 / / 解像度 : 3.9 Å Wang W / Pyle AM 資金援助 Organization Grant number 国 Howard Hughes Medical Institute (HHMI) National Institutes of Health/National Institute Of Allergy and Infectious Diseases (NIH/NIAID) R01AI131518
ジャーナル : Nat Commun / 年 : 2023タイトル : The E3 ligase Riplet promotes RIG-I signaling independent of RIG-I oligomerization.著者 : Wenshuai Wang / Benjamin Götte / Rong Guo / Anna Marie Pyle / 要旨 : RIG-I is an essential innate immune receptor that responds to infection by RNA viruses. The RIG-I signaling cascade is mediated by a series of post-translational modifications, the most important of ... RIG-I is an essential innate immune receptor that responds to infection by RNA viruses. The RIG-I signaling cascade is mediated by a series of post-translational modifications, the most important of which is ubiquitination of the RIG-I Caspase Recruitment Domains (CARDs) by E3 ligase Riplet. This is required for interaction between RIG-I and its downstream adapter protein MAVS, but the mechanism of action remains unclear. Here we show that Riplet is required for RIG-I signaling in the presence of both short and long dsRNAs, establishing that Riplet activation does not depend upon RIG-I filament formation on long dsRNAs. Likewise, quantitative Riplet-RIG-I affinity measurements establish that Riplet interacts with RIG-I regardless of whether the receptor is bound to RNA. To understand this, we solved high-resolution cryo-EM structures of RIG-I/RNA/Riplet complexes, revealing molecular interfaces that control Riplet-mediated activation and enabling the formulation of a unified model for the role of Riplet in signaling. 履歴 登録 2023年2月17日 - ヘッダ(付随情報) 公開 2023年11月15日 - マップ公開 2023年11月15日 - 更新 2024年5月29日 - 現状 2024年5月29日 処理サイト : RCSB / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報
 マップデータ
マップデータ 試料
試料 キーワード
キーワード 機能・相同性情報
機能・相同性情報 Homo sapiens (ヒト)
Homo sapiens (ヒト) データ登録者
データ登録者 米国, 2件
米国, 2件  引用
引用 ジャーナル: Nat Commun / 年: 2023
ジャーナル: Nat Commun / 年: 2023
 構造の表示
構造の表示 ダウンロードとリンク
ダウンロードとリンク emd_29825.map.gz
emd_29825.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-29825-v30.xml
emd-29825-v30.xml emd-29825.xml
emd-29825.xml EMDBヘッダ
EMDBヘッダ emd_29825_fsc.xml
emd_29825_fsc.xml FSCデータファイル
FSCデータファイル emd_29825.png
emd_29825.png emd_29825_msk_1.map
emd_29825_msk_1.map マスクマップ
マスクマップ emd-29825.cif.gz
emd-29825.cif.gz emd_29825_half_map_1.map.gz
emd_29825_half_map_1.map.gz emd_29825_half_map_2.map.gz
emd_29825_half_map_2.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-29825
http://ftp.pdbj.org/pub/emdb/structures/EMD-29825 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29825
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29825 emd_29825_validation.pdf.gz
emd_29825_validation.pdf.gz EMDB検証レポート
EMDB検証レポート emd_29825_full_validation.pdf.gz
emd_29825_full_validation.pdf.gz emd_29825_validation.xml.gz
emd_29825_validation.xml.gz emd_29825_validation.cif.gz
emd_29825_validation.cif.gz https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29825
https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29825 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29825
ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29825 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_29825.map.gz / 形式: CCP4 / 大きさ: 70.2 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_29825.map.gz / 形式: CCP4 / 大きさ: 70.2 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) emd_29825_msk_1.map
emd_29825_msk_1.map 試料の構成要素
試料の構成要素 Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト)
 Homo sapiens (ヒト)
Homo sapiens (ヒト)
 Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN
 ムービー
ムービー コントローラー
コントローラー



















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)