[English] 日本語
 Yorodumi
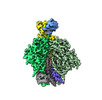
Yorodumi- EMDB-29248: Cryo-EM Structure of PG9RSH DU011 Fab in complex with BG505 DS-SO... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM Structure of PG9RSH DU011 Fab in complex with BG505 DS-SOSIP.664 | |||||||||
 Map data Map data | sharpened map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | CD4 / HIV-1 / SOSIP / Vaccine / IMMUNE SYSTEM / llama / V2 apex / therapeutic / VIRAL PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationimmunoglobulin complex / immunoglobulin mediated immune response / positive regulation of plasma membrane raft polarization / positive regulation of receptor clustering / positive regulation of establishment of T cell polarity / antigen binding / host cell endosome membrane / clathrin-dependent endocytosis of virus by host cell / blood microparticle / viral protein processing ...immunoglobulin complex / immunoglobulin mediated immune response / positive regulation of plasma membrane raft polarization / positive regulation of receptor clustering / positive regulation of establishment of T cell polarity / antigen binding / host cell endosome membrane / clathrin-dependent endocytosis of virus by host cell / blood microparticle / viral protein processing / fusion of virus membrane with host plasma membrane / virus-mediated perturbation of host defense response / fusion of virus membrane with host endosome membrane / viral envelope / virion attachment to host cell / apoptotic process / host cell plasma membrane / structural molecule activity / virion membrane / extracellular region / identical protein binding / plasma membrane Similarity search - Function | |||||||||
| Biological species |   Human immunodeficiency virus 1 / Human immunodeficiency virus 1 /  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.4 Å | |||||||||
 Authors Authors | Gorman J / Kwong PD | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Cell Rep / Year: 2023 Journal: Cell Rep / Year: 2023Title: Improved HIV-1 neutralization breadth and potency of V2-apex antibodies by in silico design. Authors: Graham T Holt / Jason Gorman / Siyu Wang / Anna U Lowegard / Baoshan Zhang / Tracy Liu / Bob C Lin / Mark K Louder / Marcel S Frenkel / Krisha McKee / Sijy O'Dell / Reda Rawi / Chen-Hsiang ...Authors: Graham T Holt / Jason Gorman / Siyu Wang / Anna U Lowegard / Baoshan Zhang / Tracy Liu / Bob C Lin / Mark K Louder / Marcel S Frenkel / Krisha McKee / Sijy O'Dell / Reda Rawi / Chen-Hsiang Shen / Nicole A Doria-Rose / Peter D Kwong / Bruce R Donald /  Abstract: Broadly neutralizing antibodies (bNAbs) against HIV can reduce viral transmission in humans, but an effective therapeutic will require unusually high breadth and potency of neutralization. We employ ...Broadly neutralizing antibodies (bNAbs) against HIV can reduce viral transmission in humans, but an effective therapeutic will require unusually high breadth and potency of neutralization. We employ the OSPREY computational protein design software to engineer variants of two apex-directed bNAbs, PGT145 and PG9RSH, resulting in increases in potency of over 100-fold against some viruses. The top designed variants improve neutralization breadth from 39% to 54% at clinically relevant concentrations (IC < 1 μg/mL) and improve median potency (IC) by up to 4-fold over a cross-clade panel of 208 strains. To investigate the mechanisms of improvement, we determine cryoelectron microscopy structures of each variant in complex with the HIV envelope trimer. Surprisingly, we find the largest increases in breadth to be a result of optimizing side-chain interactions with highly variable epitope residues. These results provide insight into mechanisms of neutralization breadth and inform strategies for antibody design and improvement. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_29248.map.gz emd_29248.map.gz | 140.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-29248-v30.xml emd-29248-v30.xml emd-29248.xml emd-29248.xml | 24.3 KB 24.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_29248.png emd_29248.png | 90.2 KB | ||
| Masks |  emd_29248_msk_1.map emd_29248_msk_1.map | 149.9 MB |  Mask map Mask map | |
| Others |  emd_29248_additional_1.map.gz emd_29248_additional_1.map.gz emd_29248_additional_2.map.gz emd_29248_additional_2.map.gz emd_29248_half_map_1.map.gz emd_29248_half_map_1.map.gz emd_29248_half_map_2.map.gz emd_29248_half_map_2.map.gz | 25.2 MB 6 MB 139.3 MB 139.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-29248 http://ftp.pdbj.org/pub/emdb/structures/EMD-29248 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29248 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29248 | HTTPS FTP |
-Validation report
| Summary document |  emd_29248_validation.pdf.gz emd_29248_validation.pdf.gz | 826.7 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_29248_full_validation.pdf.gz emd_29248_full_validation.pdf.gz | 826.2 KB | Display | |
| Data in XML |  emd_29248_validation.xml.gz emd_29248_validation.xml.gz | 14.5 KB | Display | |
| Data in CIF |  emd_29248_validation.cif.gz emd_29248_validation.cif.gz | 17.2 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29248 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29248 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29248 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29248 | HTTPS FTP |
-Related structure data
| Related structure data |  8fk5MC  8fl1C  8flwC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_29248.map.gz / Format: CCP4 / Size: 149.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_29248.map.gz / Format: CCP4 / Size: 149.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | sharpened map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.083 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_29248_msk_1.map emd_29248_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: unsharpened map
| File | emd_29248_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | unsharpened map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Resolve density modified map
| File | emd_29248_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Resolve density modified map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: half map A
| File | emd_29248_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map A | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: half map B
| File | emd_29248_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map B | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM Structure of PG9RSH DU011 Fab in complex with BG505 DS-SO...
| Entire | Name: Cryo-EM Structure of PG9RSH DU011 Fab in complex with BG505 DS-SOSIP.664 |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM Structure of PG9RSH DU011 Fab in complex with BG505 DS-SO...
| Supramolecule | Name: Cryo-EM Structure of PG9RSH DU011 Fab in complex with BG505 DS-SOSIP.664 type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#4 |
|---|---|
| Source (natural) | Organism:   Human immunodeficiency virus 1 Human immunodeficiency virus 1 |
-Macromolecule #1: Envelope glycoprotein gp41
| Macromolecule | Name: Envelope glycoprotein gp41 / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Human immunodeficiency virus 1 Human immunodeficiency virus 1 |
| Molecular weight | Theoretical: 17.146482 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: AVGIGAVFLG FLGAAGSTMG AASMTLTVQA RNLLSGIVQQ QSNLLRAPEA QQHLLKLTVW GIKQLQARVL AVERYLRDQQ LLGIWGCSG KLICCTNVPW NSSWSNRNLS EIWDNMTWLQ WDKEISNYTQ IIYGLLEESQ NQQEKNEQDL LALD UniProtKB: Envelope glycoprotein gp160 |
-Macromolecule #2: Envelope glycoprotein gp120
| Macromolecule | Name: Envelope glycoprotein gp120 / type: protein_or_peptide / ID: 2 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Human immunodeficiency virus 1 / Strain: BG505 Human immunodeficiency virus 1 / Strain: BG505 |
| Molecular weight | Theoretical: 54.086324 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: AENLWVTVYY GVPVWKDAET TLFCASDAKA YETEKHNVWA THACVPTDPN PQEIHLENVT EEFNMWKNNM VEQMHTDIIS LWDQSLKPC VKLTPLCVTL QCTNVTNNIT DDMRGELKNC SFNMTTELRD KKQKVYSLFY RLDVVQINEN QGNRSNNSNK E YRLINCNT ...String: AENLWVTVYY GVPVWKDAET TLFCASDAKA YETEKHNVWA THACVPTDPN PQEIHLENVT EEFNMWKNNM VEQMHTDIIS LWDQSLKPC VKLTPLCVTL QCTNVTNNIT DDMRGELKNC SFNMTTELRD KKQKVYSLFY RLDVVQINEN QGNRSNNSNK E YRLINCNT SACTQACPKV SFEPIPIHYC APAGFAILKC KDKKFNGTGP CPSVSTVQCT HGIKPVVSTQ LLLNGSLAEE EV MIRSENI TNNAKNILVQ FNTPVQINCT RPNNNTRKSI RIGPGQAFYA TGDIIGDIRQ AHCNVSKATW NETLGKVVKQ LRK HFGNNT IIRFANSSGG DLEVTTHSFN CGGEFFYCNT SGLFNSTWIS NTSVQGSNST GSNDSITLPC RIKQIINMWQ RIGQ CMYAP PIQGVIRCVS NITGLILTRD GGSTNSTTET FRPGGGDMRD NWRSELYKYK VVKIEPLGVA PTRCKRRVVG RRRRR R UniProtKB: Envelope glycoprotein gp160 |
-Macromolecule #3: Immunoblobulin G1 Fab heavy chain variable region (Fragment)
| Macromolecule | Name: Immunoblobulin G1 Fab heavy chain variable region (Fragment) type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 27.260258 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: ERLVESGGGV VQPGSSLRLS CAASGFDFSR QGMHWVRQAP GQGLEWVAFI KYDGSEKYHA DSVWGRLSIS RDNSKDTLYL QMNSLRVED TATYFCVREA GGPDYRNGYY (TYS)(TYS)DFYDGYYN YHYMDVWGKG TTVTVSSAST KGPSVFPLAP SSK STSGGT ...String: ERLVESGGGV VQPGSSLRLS CAASGFDFSR QGMHWVRQAP GQGLEWVAFI KYDGSEKYHA DSVWGRLSIS RDNSKDTLYL QMNSLRVED TATYFCVREA GGPDYRNGYY (TYS)(TYS)DFYDGYYN YHYMDVWGKG TTVTVSSAST KGPSVFPLAP SSK STSGGT AALGCLVKDY FPEPVTVSWN SGALTSGVHT FPAVLQSSGL YSLSSVVTVP SSSLGTQTYI CNVNHKPSNT KVDK KVEPK SCDKGLEVLF Q UniProtKB: Immunoblobulin G1 Fab heavy chain variable region |
-Macromolecule #4: Immunoglobulin lambda-1 light chain-like
| Macromolecule | Name: Immunoglobulin lambda-1 light chain-like / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 22.804182 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: QSALTQPASV SGSPGQSITI SCQGTSNDVG GYESVSWYQQ HPGKAPKVVI YDVSKRPSGV SNRFSGSKSG NTASLTISGL QAEDEGDYY CKSLTSRSHR VFGTGTKLTV LGQPKAAPSV TLFPPSSEEL QANKATLVCL ISDFYPGAVT VAWKADSSPV K AGVETTTP ...String: QSALTQPASV SGSPGQSITI SCQGTSNDVG GYESVSWYQQ HPGKAPKVVI YDVSKRPSGV SNRFSGSKSG NTASLTISGL QAEDEGDYY CKSLTSRSHR VFGTGTKLTV LGQPKAAPSV TLFPPSSEEL QANKATLVCL ISDFYPGAVT VAWKADSSPV K AGVETTTP SKQSNNKYAA SSYLSLTPEQ WKSHKSYSCQ VTHEGSTVEK TVAPTECS UniProtKB: Ig-like domain-containing protein |
-Macromolecule #9: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 9 / Number of copies: 38 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2 mg/mL |
|---|---|
| Buffer | pH: 7.4 / Details: PBS |
| Grid | Model: C-flat-1.2/1.3 / Support film - Material: CARBON / Support film - topology: HOLEY |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 90 % / Chamber temperature: 293 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Detector mode: COUNTING / Number grids imaged: 1 / Average exposure time: 2.0 sec. / Average electron dose: 63.75 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)