登録情報 データベース : EMDB / ID : EMD-26828タイトル Citrus V-ATPase State 2, H in contact with subunit a Citrus V-ATPase State 2, H in contact with subunit a / / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
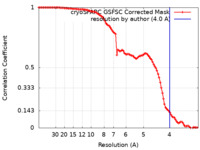
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Citrus limon (植物)手法 / / 解像度 : 4.0 Å Tan YZ / Rubinstein JL 資金援助 Organization Grant number 国 Natural Sciences and Engineering Research Council (NSERC, Canada)
ジャーナル : Structure / 年 : 2022タイトル : Structure of V-ATPase from citrus fruit.著者 : Yong Zi Tan / Kristine A Keon / Rana Abdelaziz / Peter Imming / Waltraud Schulze / Karin Schumacher / John L Rubinstein / 要旨 : We used the Legionella pneumophila effector SidK to affinity purify the endogenous vacuolar-type ATPases (V-ATPases) from lemon fruit. The preparation was sufficient for cryoelectron microscopy, ... We used the Legionella pneumophila effector SidK to affinity purify the endogenous vacuolar-type ATPases (V-ATPases) from lemon fruit. The preparation was sufficient for cryoelectron microscopy, allowing structure determination of the enzyme in two rotational states. The structure defines the ATP:H ratio of the enzyme, demonstrating that it can establish a maximum ΔpH of ∼3, which is insufficient to maintain the low pH observed in the vacuoles of juice sac cells in lemons and other citrus fruit. Compared with yeast and mammalian enzymes, the membrane region of the plant V-ATPase lacks subunit f and possesses an unusual configuration of transmembrane α helices. Subunit H, which inhibits ATP hydrolysis in the isolated catalytic region of V-ATPase, adopts two different conformations in the intact complex, hinting at a role in modulating activity in the intact enzyme. 履歴 登録 2022年5月3日 - ヘッダ(付随情報) 公開 2022年7月6日 - マップ公開 2022年7月6日 - 更新 2024年1月17日 - 現状 2024年1月17日 処理サイト : RCSB / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報
 マップデータ
マップデータ 試料
試料 キーワード
キーワード 機能・相同性情報
機能・相同性情報
 データ登録者
データ登録者 カナダ, 1件
カナダ, 1件  引用
引用 ジャーナル: Structure / 年: 2022
ジャーナル: Structure / 年: 2022

 構造の表示
構造の表示 ダウンロードとリンク
ダウンロードとリンク emd_26828.map.gz
emd_26828.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-26828-v30.xml
emd-26828-v30.xml emd-26828.xml
emd-26828.xml EMDBヘッダ
EMDBヘッダ emd_26828_fsc.xml
emd_26828_fsc.xml FSCデータファイル
FSCデータファイル emd_26828.png
emd_26828.png emd-26828.cif.gz
emd-26828.cif.gz emd_26828_additional_1.map.gz
emd_26828_additional_1.map.gz emd_26828_half_map_1.map.gz
emd_26828_half_map_1.map.gz emd_26828_half_map_2.map.gz
emd_26828_half_map_2.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-26828
http://ftp.pdbj.org/pub/emdb/structures/EMD-26828 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26828
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26828 emd_26828_validation.pdf.gz
emd_26828_validation.pdf.gz EMDB検証レポート
EMDB検証レポート emd_26828_full_validation.pdf.gz
emd_26828_full_validation.pdf.gz emd_26828_validation.xml.gz
emd_26828_validation.xml.gz emd_26828_validation.cif.gz
emd_26828_validation.cif.gz https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26828
https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26828 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26828
ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26828 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_26828.map.gz / 形式: CCP4 / 大きさ: 103 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_26828.map.gz / 形式: CCP4 / 大きさ: 103 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) 試料の構成要素
試料の構成要素 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN
 ムービー
ムービー コントローラー
コントローラー

















 Z
Z Y
Y X
X