+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-22650 | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of STRIPAK complex | ||||||||||||||||||
 Map data Map data | SK7 primary map | ||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||
 Keywords Keywords | phosphorylation / complex / PP2A / SIGNALING PROTEIN | ||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationarmadillo repeat domain binding / meiotic spindle elongation / Integration of energy metabolism / PP2A-mediated dephosphorylation of key metabolic factors / regulation of microtubule binding / protein serine/threonine phosphatase complex / mitotic sister chromatid separation / MASTL Facilitates Mitotic Progression / regulation of meiotic cell cycle process involved in oocyte maturation / protein phosphatase type 2A complex ...armadillo repeat domain binding / meiotic spindle elongation / Integration of energy metabolism / PP2A-mediated dephosphorylation of key metabolic factors / regulation of microtubule binding / protein serine/threonine phosphatase complex / mitotic sister chromatid separation / MASTL Facilitates Mitotic Progression / regulation of meiotic cell cycle process involved in oocyte maturation / protein phosphatase type 2A complex / meiotic sister chromatid cohesion, centromeric / peptidyl-serine dephosphorylation / negative regulation of intracellular estrogen receptor signaling pathway / peptidyl-threonine dephosphorylation / FAR/SIN/STRIPAK complex / positive regulation of microtubule binding / Regulation of glycolysis by fructose 2,6-bisphosphate metabolism / Inhibition of replication initiation of damaged DNA by RB1/E2F1 / female meiotic nuclear division / protein phosphatase regulator activity / protein antigen binding / GABA receptor binding / APC truncation mutants have impaired AXIN binding / AXIN missense mutants destabilize the destruction complex / Truncations of AMER1 destabilize the destruction complex / Initiation of Nuclear Envelope (NE) Reformation / ERKs are inactivated / positive regulation of extrinsic apoptotic signaling pathway in absence of ligand / Beta-catenin phosphorylation cascade / Signaling by GSK3beta mutants / CTNNB1 S33 mutants aren't phosphorylated / CTNNB1 S37 mutants aren't phosphorylated / CTNNB1 S45 mutants aren't phosphorylated / CTNNB1 T41 mutants aren't phosphorylated / negative regulation of epithelial to mesenchymal transition / regulation of cell morphogenesis / regulation of growth / Disassembly of the destruction complex and recruitment of AXIN to the membrane / cortical actin cytoskeleton organization / negative regulation of glycolytic process through fructose-6-phosphate / positive regulation of NLRP3 inflammasome complex assembly / myosin phosphatase activity / Golgi cisterna membrane / CTLA4 inhibitory signaling / protein serine/threonine phosphatase activity / Platelet sensitization by LDL / protein-serine/threonine phosphatase / Cyclin A/B1/B2 associated events during G2/M transition / regulation of cell differentiation / negative regulation of hippo signaling / ERK/MAPK targets / T cell homeostasis / regulation of G1/S transition of mitotic cell cycle / mesoderm development / phosphoprotein phosphatase activity / chromosome, centromeric region / negative regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / DARPP-32 events / lateral plasma membrane / meiotic cell cycle / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / Mitotic Prometaphase / EML4 and NUDC in mitotic spindle formation / Loss of Nlp from mitotic centrosomes / Loss of proteins required for interphase microtubule organization from the centrosome / Recruitment of mitotic centrosome proteins and complexes / cytoskeleton organization / Recruitment of NuMA to mitotic centrosomes / Anchoring of the basal body to the plasma membrane / Resolution of Sister Chromatid Cohesion / protein dephosphorylation / AURKA Activation by TPX2 / protein phosphatase 2A binding / protein tyrosine phosphatase activity / chromosome segregation / RHO GTPases Activate Formins / kinase binding / response to lead ion / RAF activation / regulation of protein phosphorylation / Spry regulation of FGF signaling / Degradation of beta-catenin by the destruction complex / tau protein binding / positive regulation of protein serine/threonine kinase activity / PKR-mediated signaling / Cyclin D associated events in G1 / small GTPase binding / spindle pole / Regulation of TP53 Degradation / Negative regulation of MAPK pathway / Separation of Sister Chromatids / microtubule cytoskeleton / Regulation of PLK1 Activity at G2/M Transition / response to estradiol / mitotic cell cycle / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling / protein-macromolecule adaptor activity / protein-containing complex assembly / dendritic spine Similarity search - Function | ||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.3 Å | ||||||||||||||||||
 Authors Authors | Jeong B-C / Bai XC | ||||||||||||||||||
| Funding support |  United States, 5 items United States, 5 items
| ||||||||||||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2021 Journal: Nat Struct Mol Biol / Year: 2021Title: Cryo-EM structure of the Hippo signaling integrator human STRIPAK. Authors: Byung-Cheon Jeong / Sung Jun Bae / Lisheng Ni / Xuewu Zhang / Xiao-Chen Bai / Xuelian Luo /  Abstract: The striatin-interacting phosphatase and kinase (STRIPAK) complex is a large, multisubunit protein phosphatase 2A (PP2A) assembly that integrates diverse cellular signals in the Hippo pathway to ...The striatin-interacting phosphatase and kinase (STRIPAK) complex is a large, multisubunit protein phosphatase 2A (PP2A) assembly that integrates diverse cellular signals in the Hippo pathway to regulate cell proliferation and survival. The architecture and assembly mechanism of this critical complex are poorly understood. Using cryo-EM, we determine the structure of the human STRIPAK core comprising PP2AA, PP2AC, STRN3, STRIP1, and MOB4 at 3.2-Å resolution. Unlike the canonical trimeric PP2A holoenzyme, STRIPAK contains four copies of STRN3 and one copy of each the PP2AA-C heterodimer, STRIP1, and MOB4. The STRN3 coiled-coil domains form an elongated homotetrameric scaffold that links the complex together. An inositol hexakisphosphate (IP) is identified as a structural cofactor of STRIP1. Mutations of key residues at subunit interfaces disrupt the integrity of STRIPAK, causing aberrant Hippo pathway activation. Thus, STRIPAK is established as a noncanonical PP2A complex with four copies of regulatory STRN3 for enhanced signal integration. | ||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_22650.map.gz emd_22650.map.gz | 166.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-22650-v30.xml emd-22650-v30.xml emd-22650.xml emd-22650.xml | 25.4 KB 25.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_22650.png emd_22650.png | 348.2 KB | ||
| Filedesc metadata |  emd-22650.cif.gz emd-22650.cif.gz | 8.3 KB | ||
| Others |  emd_22650_additional_1.map.gz emd_22650_additional_1.map.gz | 166.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-22650 http://ftp.pdbj.org/pub/emdb/structures/EMD-22650 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-22650 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-22650 | HTTPS FTP |
-Related structure data
| Related structure data |  7k36MC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_22650.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_22650.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | SK7 primary map | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.83 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: Focused map
| File | emd_22650_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Focused map | ||||||||||||
| Projections & Slices |
| ||||||||||||
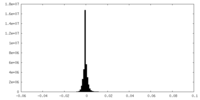
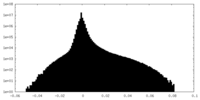
| Density Histograms |
- Sample components
Sample components
-Entire : SK7 complex
| Entire | Name: SK7 complex |
|---|---|
| Components |
|
-Supramolecule #1: SK7 complex
| Supramolecule | Name: SK7 complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 817.8 KDa |
-Macromolecule #1: Serine/threonine-protein phosphatase 2A 65 kDa regulatory subunit...
| Macromolecule | Name: Serine/threonine-protein phosphatase 2A 65 kDa regulatory subunit A alpha isoform type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 65.378344 KDa |
| Recombinant expression | Organism:  Baculovirus expression vector pFastBac1-HM Baculovirus expression vector pFastBac1-HM |
| Sequence | String: MAAADGDDSL YPIAVLIDEL RNEDVQLRLN SIKKLSTIAL ALGVERTRSE LLPFLTDTIY DEDEVLLALA EQLGTFTTLV GGPEYVHCL LPPLESLATV EETVVRDKAV ESLRAISHEH SPSDLEAHFV PLVKRLAGGD WFTSRTSACG LFSVCYPRVS S AVKAELRQ ...String: MAAADGDDSL YPIAVLIDEL RNEDVQLRLN SIKKLSTIAL ALGVERTRSE LLPFLTDTIY DEDEVLLALA EQLGTFTTLV GGPEYVHCL LPPLESLATV EETVVRDKAV ESLRAISHEH SPSDLEAHFV PLVKRLAGGD WFTSRTSACG LFSVCYPRVS S AVKAELRQ YFRNLCSDDT PMVRRAAASK LGEFAKVLEL DNVKSEIIPM FSNLASDEQD SVRLLAVEAC VNIAQLLPQE DL EALVMPT LRQAAEDKSW RVRYMVADKF TELQKAVGPE ITKTDLVPAF QNLMKDCEAE VRAAASHKVK EFCENLSADC REN VIMSQI LPCIKELVSD ANQHVKSALA SVIMGLSPIL GKDNTIEHLL PLFLAQLKDE CPEVRLNIIS NLDCVNEVIG IRQL SQSLL PAIVELAEDA KWRVRLAIIE YMPLLAGQLG VEFFDEKLNS LCMAWLVDHV YAIREAATSN LKKLVEKFGK EWAHA TIIP KVLAMSGDPN YLHRMTTLFC INVLSEVCGQ DITTKHMLPT VLRMAGDPVA NVRFNVAKSL QKIGPILDNS TLQSEV KPI LEKLTQDQDV DVKYFAQEAL TVLSLA UniProtKB: Serine/threonine-protein phosphatase 2A 65 kDa regulatory subunit A alpha isoform |
-Macromolecule #2: Striatin-3
| Macromolecule | Name: Striatin-3 / type: protein_or_peptide / ID: 2 / Number of copies: 5 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 77.833484 KDa |
| Recombinant expression | Organism:  Baculovirus expression vector pFastBac1-HM Baculovirus expression vector pFastBac1-HM |
| Sequence | String: MDELAGGGGG GPGMAAPPRQ QQGPGGNLGL SPGGNGAAGG GGPPASEGAG PAAGPELSRP QQYTIPGILH YIQHEWARFE MERAHWEVE RAELQARIAF LQGERKGQEN LKKDLVRRIK MLEYALKQER AKYHKLKYGT ELNQGDLKMP TFESEETKDT E APTAPQNS ...String: MDELAGGGGG GPGMAAPPRQ QQGPGGNLGL SPGGNGAAGG GGPPASEGAG PAAGPELSRP QQYTIPGILH YIQHEWARFE MERAHWEVE RAELQARIAF LQGERKGQEN LKKDLVRRIK MLEYALKQER AKYHKLKYGT ELNQGDLKMP TFESEETKDT E APTAPQNS QLTWKQGRQL LRQYLQEVGY TDTILDVRSQ RVRSLLGLSN SEPNGSVETK NLEQILNGGE SPKQKGQEIK RS SGDVLET FNFLENADDS DEDEENDMIE GIPEGKDKHR MNKHKIGNEG LAADLTDDPD TEEALKEFDF LVTAEDGEGA GEA RSSGDG TEWAEPITFP SGGGKSFIMG SDDVLLSVLG LGDLADLTVT NDADYSYDLP ANKDAFRKTW NPKYTLRSHF DGVR ALAFH PVEPVLVTAS EDHTLKLWNL QKTVPAKKSA SLDVEPIYTF RAHIGPVLSL AISSNGEQCF SGGIDATIQW WNMPS PSVD PYDTYEPNVL AGTLVGHTDA VWGLAYSGIK NQLLSCSADG TVRLWNPQEK LPCICTYNGD KKHGIPTSVD FIGCDP AHM VTSFNTGSAV IYDLETSQSL VILSSQVDSG LQSNNHINRV VSHPTLPVTI TAHEDRHIKF FDNKTGKMIH SMVAHLD AV TSLAVDPNGI YLMSGSHDCS IRLWNLDSKT CVQEITAHRK KLDESIYDVA FHSSKAYIAS AGADALAKVF V UniProtKB: Striatin-3 |
-Macromolecule #3: Serine/threonine-protein phosphatase 2A catalytic subunit alpha i...
| Macromolecule | Name: Serine/threonine-protein phosphatase 2A catalytic subunit alpha isoform type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO / EC number: protein-serine/threonine phosphatase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 35.636152 KDa |
| Recombinant expression | Organism:  Baculovirus expression vector pFastBac1-HM Baculovirus expression vector pFastBac1-HM |
| Sequence | String: MDEKVFTKEL DQWIEQLNEC KQLSESQVKS LCEKAKEILT KESNVQEVRC PVTVCGDVHG QFHDLMELFR IGGKSPDTNY LFMGDYVDR GYYSVETVTL LVALKVRYRE RITILRGNHE SRQITQVYGF YDECLRKYGN ANVWKYFTDL FDYLPLTALV D GQIFCLHG ...String: MDEKVFTKEL DQWIEQLNEC KQLSESQVKS LCEKAKEILT KESNVQEVRC PVTVCGDVHG QFHDLMELFR IGGKSPDTNY LFMGDYVDR GYYSVETVTL LVALKVRYRE RITILRGNHE SRQITQVYGF YDECLRKYGN ANVWKYFTDL FDYLPLTALV D GQIFCLHG GLSPSIDTLD HIRALDRLQE VPHEGPMCDL LWSDPDDRGG WGISPRGAGY TFGQDISETF NHANGLTLVS RA HQLVMEG YNWCHDRNVV TIFSAPNYCY RCGNQAAIME LDDTLKYSFL QFDPAPRRGE PHVTRRTPDY FL UniProtKB: Serine/threonine-protein phosphatase 2A catalytic subunit alpha isoform |
-Macromolecule #4: MOB-like protein phocein
| Macromolecule | Name: MOB-like protein phocein / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 26.064447 KDa |
| Recombinant expression | Organism:  Baculovirus expression vector pFastBac1-HM Baculovirus expression vector pFastBac1-HM |
| Sequence | String: MVMAEGTAVL RRNRPGTKAQ DFYNWPDESF DEMDSTLAVQ QYIQQNIRAD CSNIDKILEP PEGQDEGVWK YEHLRQFCLE LNGLAVKLQ SECHPDTCTQ MTATEQWIFL CAAHKTPKEC PAIDYTRHTL DGAACLLNSN KYFPSRVSIK ESSVAKLGSV C RRIYRIFS ...String: MVMAEGTAVL RRNRPGTKAQ DFYNWPDESF DEMDSTLAVQ QYIQQNIRAD CSNIDKILEP PEGQDEGVWK YEHLRQFCLE LNGLAVKLQ SECHPDTCTQ MTATEQWIFL CAAHKTPKEC PAIDYTRHTL DGAACLLNSN KYFPSRVSIK ESSVAKLGSV C RRIYRIFS HAYFHHRQIF DEYENETFLC HRFTKFVMKY NLMSKDNLIV PILEEEVQNS VSGESEA UniProtKB: MOB-like protein phocein |
-Macromolecule #5: Striatin-interacting protein 1
| Macromolecule | Name: Striatin-interacting protein 1 / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 95.695656 KDa |
| Recombinant expression | Organism:  Baculovirus expression vector pFastBac1-HM Baculovirus expression vector pFastBac1-HM |
| Sequence | String: MEPAVGGPGP LIVNNKQPQP PPPPPPAAAQ PPPGAPRAAA GLLPGGKARE FNRNQRKDSE GYSESPDLEF EYADTDKWAA ELSELYSYT EGPEFLMNRK CFEEDFRIHV TDKKWTELDT NQHRTHAMRL LDGLEVTARE KRLKVARAIL YVAQGTFGEC S SEAEVQSW ...String: MEPAVGGPGP LIVNNKQPQP PPPPPPAAAQ PPPGAPRAAA GLLPGGKARE FNRNQRKDSE GYSESPDLEF EYADTDKWAA ELSELYSYT EGPEFLMNRK CFEEDFRIHV TDKKWTELDT NQHRTHAMRL LDGLEVTARE KRLKVARAIL YVAQGTFGEC S SEAEVQSW MRYNIFLLLE VGTFNALVEL LNMEIDNSAA CSSAVRKPAI SLADSTDLRV LLNIMYLIVE TVHQECEGDK AE WRTMRQT FRAELGSPLY NNEPFAIMLF GMVTKFCSGH APHFPMKKVL LLLWKTVLCT LGGFEELQSM KAEKRSILGL PPL PEDSIK VIRNMRAASP PASASDLIEQ QQKRGRREHK ALIKQDNLDA FNERDPYKAD DSREEEEEND DDNSLEGETF PLER DEVMP PPLQHPQTDR LTCPKGLPWA PKVREKDIEM FLESSRSKFI GYTLGSDTNT VVGLPRPIHE SIKTLKQHKY TSIAE VQAQ MEEEYLRSPL SGGEEEVEQV PAETLYQGLL PSLPQYMIAL LKILLAAAPT SKAKTDSINI LADVLPEEMP TTVLQS MKL GVDVNRHKEV IVKAISAVLL LLLKHFKLNH VYQFEYMAQH LVFANCIPLI LKFFNQNIMS YITAKNSISV LDYPHCV VH ELPELTAESL EAGDSNQFCW RNLFSCINLL RILNKLTKWK HSRTMMLVVF KSAPILKRAL KVKQAMMQLY VLKLLKVQ T KYLGRQWRKS NMKTMSAIYQ KVRHRLNDDW AYGNDLDARP WDFQAEECAL RANIERFNAR RYDRAHSNPD FLPVDNCLQ SVLGQRVDLP EDFQMNYDLW LEREVFSKPI SWEELLQ UniProtKB: Striatin-interacting protein 1 |
-Macromolecule #6: MANGANESE (II) ION
| Macromolecule | Name: MANGANESE (II) ION / type: ligand / ID: 6 / Number of copies: 2 / Formula: MN |
|---|---|
| Molecular weight | Theoretical: 54.938 Da |
-Macromolecule #7: ZINC ION
| Macromolecule | Name: ZINC ION / type: ligand / ID: 7 / Number of copies: 2 / Formula: ZN |
|---|---|
| Molecular weight | Theoretical: 65.409 Da |
-Macromolecule #8: INOSITOL HEXAKISPHOSPHATE
| Macromolecule | Name: INOSITOL HEXAKISPHOSPHATE / type: ligand / ID: 8 / Number of copies: 1 / Formula: IHP |
|---|---|
| Molecular weight | Theoretical: 660.035 Da |
| Chemical component information |  ChemComp-IHP: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2.0 mg/mL | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
| ||||||||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 80 sec. / Pretreatment - Atmosphere: AIR / Details: The grid was coated with gold prior to use | ||||||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Detector mode: SUPER-RESOLUTION / Number real images: 4356 / Average electron dose: 64.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Calibrated magnification: 59524 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model |
| ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Space: REAL / Protocol: AB INITIO MODEL | ||||||||||
| Output model |  PDB-7k36: |
 Movie
Movie Controller
Controller




























 Z
Z Y
Y X
X