+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | 2.7 A cryo-EM structure of in vitro assembled type 1 pilus rod | |||||||||||||||
 Map data Map data | sharpened map | |||||||||||||||
 Sample Sample |
| |||||||||||||||
 Keywords Keywords | FimA / pilus / monomer / subunit / pili / main structural subunit / high resolution / STRUCTURAL PROTEIN / cryo-EM / helical processing / RELION / Chaperone-usher pilus | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationcell adhesion involved in single-species biofilm formation / pilus / cell adhesion / identical protein binding Similarity search - Function | |||||||||||||||
| Biological species |  | |||||||||||||||
| Method | helical reconstruction / cryo EM / Resolution: 2.7 Å | |||||||||||||||
 Authors Authors | Hospenthal M / Zyla D / Glockshuber R / Waksman G | |||||||||||||||
| Funding support |  Switzerland, Switzerland,  United Kingdom, 4 items United Kingdom, 4 items
| |||||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2024 Journal: Nat Commun / Year: 2024Title: The assembly platform FimD is required to obtain the most stable quaternary structure of type 1 pili. Authors: Dawid S Zyla / Thomas Wiegand / Paul Bachmann / Rafal Zdanowicz / Christoph Giese / Beat H Meier / Gabriel Waksman / Manuela K Hospenthal / Rudi Glockshuber /     Abstract: Type 1 pili are important virulence factors of uropathogenic Escherichia coli that mediate bacterial attachment to epithelial cells in the urinary tract. The pilus rod is comprised of thousands of ...Type 1 pili are important virulence factors of uropathogenic Escherichia coli that mediate bacterial attachment to epithelial cells in the urinary tract. The pilus rod is comprised of thousands of copies of the main structural subunit FimA and is assembled in vivo by the assembly platform FimD. Although type 1 pilus rods can self-assemble from FimA in vitro, this reaction is slower and produces structures with lower kinetic stability against denaturants compared to in vivo-assembled rods. Our study reveals that FimD-catalysed in vitro-assembled type 1 pilus rods attain a similar stability as pilus rods assembled in vivo. Employing structural, biophysical and biochemical analyses, we show that in vitro assembly reactions lacking FimD produce pilus rods with structural defects, reducing their stability against dissociation. Overall, our results indicate that FimD is not only required for the catalysis of pilus assembly, but also to control the assembly of the most stable quaternary structure. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_17863.map.gz emd_17863.map.gz | 4.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-17863-v30.xml emd-17863-v30.xml emd-17863.xml emd-17863.xml | 17.8 KB 17.8 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_17863.png emd_17863.png | 54.3 KB | ||
| Masks |  emd_17863_msk_1.map emd_17863_msk_1.map | 59.6 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-17863.cif.gz emd-17863.cif.gz | 6.1 KB | ||
| Others |  emd_17863_half_map_1.map.gz emd_17863_half_map_1.map.gz emd_17863_half_map_2.map.gz emd_17863_half_map_2.map.gz | 46.2 MB 46.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17863 http://ftp.pdbj.org/pub/emdb/structures/EMD-17863 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17863 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17863 | HTTPS FTP |
-Validation report
| Summary document |  emd_17863_validation.pdf.gz emd_17863_validation.pdf.gz | 849.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_17863_full_validation.pdf.gz emd_17863_full_validation.pdf.gz | 848.9 KB | Display | |
| Data in XML |  emd_17863_validation.xml.gz emd_17863_validation.xml.gz | 12.1 KB | Display | |
| Data in CIF |  emd_17863_validation.cif.gz emd_17863_validation.cif.gz | 14.2 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17863 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17863 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17863 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17863 | HTTPS FTP |
-Related structure data
| Related structure data |  8psvMC  6y7sC  8ptuC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_17863.map.gz / Format: CCP4 / Size: 59.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_17863.map.gz / Format: CCP4 / Size: 59.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | sharpened map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.055 Å | ||||||||||||||||||||||||||||||||||||
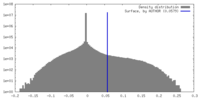
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_17863_msk_1.map emd_17863_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: halfmap1
| File | emd_17863_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | halfmap1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: halfmap2
| File | emd_17863_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | halfmap2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Type 1 pilus rod
| Entire | Name: Type 1 pilus rod |
|---|---|
| Components |
|
-Supramolecule #1: Type 1 pilus rod
| Supramolecule | Name: Type 1 pilus rod / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all Details: Type 1 pili assembled in vitro from recombinantly expressed as inclusion bodies and refolded FimA. |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 20.3 kDa/nm |
-Macromolecule #1: Type-1 fimbrial protein, A chain
| Macromolecule | Name: Type-1 fimbrial protein, A chain / type: protein_or_peptide / ID: 1 / Number of copies: 6 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 18.121074 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MKIKTLAIVV LSALSLSSTA ALAAATTVNG GTVHFKGEVV NAACAVDAGS VDQTVQLGQV RTASLAQEGA TSSAVGFNIQ LNDCDTNVA SKAAVAFLGT AIDAGHTNVL ALQSSAAGSA TNVGVQILDR TGAALTLDGA TFSSETTLNN GTNTIPFQAR Y FATGAATP GAANADATFK VQYQ UniProtKB: Type-1 fimbrial protein, A chain |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Concentration | 1.58 mg/mL |
|---|---|
| Buffer | pH: 7 / Details: in ddH2O. |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 70 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV Details: 3 ul sample, 30 s wait time, 0.5 s drain time, 6 s blotting. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Digitization - Dimensions - Width: 8000 pixel / Digitization - Dimensions - Height: 8000 pixel / Number grids imaged: 1 / Average exposure time: 14.0 sec. / Average electron dose: 97.54 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.5 µm / Nominal defocus min: 1.5 µm / Nominal magnification: 94340 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Applied symmetry - Helical parameters - Δz: 7.76 Å Applied symmetry - Helical parameters - Δ&Phi: 115.0 ° Applied symmetry - Helical parameters - Axial symmetry: C1 (asymmetric) Resolution.type: BY AUTHOR / Resolution: 2.7 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION (ver. 3.08) / Number images used: 11000 |
|---|---|
| Segment selection | Number selected: 516000 Details: Autopicking based on the 2D classes from manually chosen filaments |
| Startup model | Type of model: OTHER / Details: featureless cylinder |
| Final angle assignment | Type: NOT APPLICABLE / Software - Name: RELION (ver. 3.08) |
-Atomic model buiding 1
| Initial model | PDB ID: Chain - Chain ID: D / Chain - Source name: PDB / Chain - Initial model type: experimental model |
|---|---|
| Refinement | Space: REAL / Protocol: RIGID BODY FIT / Overall B value: 45.07 / Target criteria: Correlation coefficient |
| Output model |  PDB-8psv: |
 Movie
Movie Controller
Controller






 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)