[English] 日本語
 Yorodumi
Yorodumi- EMDB-17794: HK97 small terminase in complex with DNA after focused classification -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | HK97 small terminase in complex with DNA after focused classification | |||||||||
 Map data Map data | HK97 small terminase | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | small terminase / HK97 / bacteriophage / complex with DNA / DNA BINDING PROTEIN | |||||||||
| Function / homology | Terminase small subunit Function and homology information Function and homology information | |||||||||
| Biological species |  Hendrixvirus / Hendrixvirus /  Escherichia phage HK97 (virus) / Escherichia phage HK97 (virus) /  Byrnievirus HK97 Byrnievirus HK97 | |||||||||
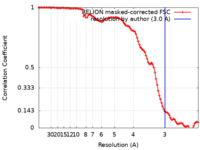
| Method | single particle reconstruction / cryo EM / Resolution: 3.0 Å | |||||||||
 Authors Authors | Chechik M / Greive SJ / Antson AA / Jenkins HT | |||||||||
| Funding support |  United Kingdom, 1 items United Kingdom, 1 items
| |||||||||
 Citation Citation | Journal: bioRxiv / Year: 2023 Title: Structure of HK97 small terminase:DNA complex unveils a novel DNA binding mechanism by a circular protein. Authors: Maria Chechik / Sandra J Greive / Alfred A Antson / Huw T Jenkins /  Abstract: DNA recognition is critical for assembly of double-stranded DNA viruses, in particular for the initiation of packaging the viral genome into the capsid. DNA packaging has been extensively studied for ...DNA recognition is critical for assembly of double-stranded DNA viruses, in particular for the initiation of packaging the viral genome into the capsid. DNA packaging has been extensively studied for three archetypal bacteriophage systems: , and phi29. We identified the minimal site within the region of bacteriophage HK97 specifically recognised by the small terminase and determined a cryoEM structure for the small terminase:DNA complex. This nonameric circular protein utilizes a previously unknown mechanism of DNA binding. While DNA threads through the central tunnel, unexpectedly, DNA-recognition is generated at its exit by a substructure formed by the N- and C-terminal segments of two adjacent protomers of the terminase which are unstructured in the absence of DNA. Such interaction ensures continuous engagement of the small terminase with DNA, allowing sliding along DNA while simultaneously checking the DNA sequence. This mechanism allows locating and instigating packaging initiation and termination precisely at the site. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_17794.map.gz emd_17794.map.gz | 7.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-17794-v30.xml emd-17794-v30.xml emd-17794.xml emd-17794.xml | 21.4 KB 21.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_17794_fsc.xml emd_17794_fsc.xml | 10.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_17794.png emd_17794.png | 36.1 KB | ||
| Masks |  emd_17794_msk_1.map emd_17794_msk_1.map | 8 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-17794.cif.gz emd-17794.cif.gz | 6.6 KB | ||
| Others |  emd_17794_half_map_1.map.gz emd_17794_half_map_1.map.gz emd_17794_half_map_2.map.gz emd_17794_half_map_2.map.gz | 7.4 MB 7.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17794 http://ftp.pdbj.org/pub/emdb/structures/EMD-17794 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17794 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17794 | HTTPS FTP |
-Validation report
| Summary document |  emd_17794_validation.pdf.gz emd_17794_validation.pdf.gz | 1022 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_17794_full_validation.pdf.gz emd_17794_full_validation.pdf.gz | 1021.6 KB | Display | |
| Data in XML |  emd_17794_validation.xml.gz emd_17794_validation.xml.gz | 13.2 KB | Display | |
| Data in CIF |  emd_17794_validation.cif.gz emd_17794_validation.cif.gz | 17.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17794 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17794 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17794 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17794 | HTTPS FTP |
-Related structure data
| Related structure data |  8popMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_17794.map.gz / Format: CCP4 / Size: 8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_17794.map.gz / Format: CCP4 / Size: 8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | HK97 small terminase | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.18889 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_17794_msk_1.map emd_17794_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: HK97 small terminase
| File | emd_17794_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | HK97 small terminase | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: HK97 small terminase
| File | emd_17794_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | HK97 small terminase | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : HK97 small terminase in complex with DNA
| Entire | Name: HK97 small terminase in complex with DNA |
|---|---|
| Components |
|
-Supramolecule #1: HK97 small terminase in complex with DNA
| Supramolecule | Name: HK97 small terminase in complex with DNA / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Hendrixvirus Hendrixvirus |
-Macromolecule #1: Terminase small subunit
| Macromolecule | Name: Terminase small subunit / type: protein_or_peptide / ID: 1 / Number of copies: 9 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Escherichia phage HK97 (virus) Escherichia phage HK97 (virus) |
| Molecular weight | Theoretical: 18.497648 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MADKRIRSDS SAAAVQAMKN AAVDTIDPPS HAGLEKKAEP FWHDNIRSKA LDSWTPADLL AAVELANNQL YITVLRKDLR KEERIRGEE RDEGLIKDLR KQIVELQRTI LAQRRDLQIH SHATNGESRD QKKRNQNDRD ARNTKNEHQD QDDNLIAFPK H G UniProtKB: Terminase small subunit |
-Macromolecule #2: DNA (31-MER)
| Macromolecule | Name: DNA (31-MER) / type: dna / ID: 2 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:  Byrnievirus HK97 Byrnievirus HK97 |
| Molecular weight | Theoretical: 9.600261 KDa |
| Sequence | String: (DT)(DA)(DA)(DA)(DA)(DC)(DT)(DA)(DA)(DA) (DA)(DA)(DA)(DA)(DT)(DC)(DG)(DG)(DG)(DT) (DT)(DA)(DG)(DC)(DG)(DT)(DT)(DA)(DA) (DA)(DT) |
-Macromolecule #3: DNA (31-MER)
| Macromolecule | Name: DNA (31-MER) / type: dna / ID: 3 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:  Byrnievirus HK97 Byrnievirus HK97 |
| Molecular weight | Theoretical: 9.457115 KDa |
| Sequence | String: (DA)(DT)(DT)(DT)(DA)(DA)(DC)(DG)(DC)(DT) (DA)(DA)(DC)(DC)(DC)(DG)(DA)(DT)(DT)(DT) (DT)(DT)(DT)(DT)(DA)(DG)(DT)(DT)(DT) (DT)(DA) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 Component:
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grid | Model: UltrAuFoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 180 sec. / Pretreatment - Atmosphere: AIR / Pretreatment - Pressure: 0.038 kPa | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV | ||||||||||||
| Details | complex was purified on S200 10/30 column |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Number grids imaged: 1 / Number real images: 2767 / Average exposure time: 8.0 sec. / Average electron dose: 53.6 e/Å2 Details: Two datasets were collected from the same grid. 1st set: 682 images 2nd set: 2085 images |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.4 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 130000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)