+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Cryo-EM structure of the wild-type alpha-synuclein fibril. | |||||||||
 マップデータ マップデータ | Cryo-EM structure of the wild-type alpha-synuclein fibril. | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | alpha-synuclein / amyloid / fibril / Parkinson's disease / PROTEIN FIBRIL | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報negative regulation of mitochondrial electron transport, NADH to ubiquinone / neutral lipid metabolic process / regulation of phospholipase activity / negative regulation of monooxygenase activity / regulation of acyl-CoA biosynthetic process / negative regulation of dopamine uptake involved in synaptic transmission / negative regulation of norepinephrine uptake / positive regulation of glutathione peroxidase activity / positive regulation of SNARE complex assembly / positive regulation of hydrogen peroxide catabolic process ...negative regulation of mitochondrial electron transport, NADH to ubiquinone / neutral lipid metabolic process / regulation of phospholipase activity / negative regulation of monooxygenase activity / regulation of acyl-CoA biosynthetic process / negative regulation of dopamine uptake involved in synaptic transmission / negative regulation of norepinephrine uptake / positive regulation of glutathione peroxidase activity / positive regulation of SNARE complex assembly / positive regulation of hydrogen peroxide catabolic process / supramolecular fiber / negative regulation of transporter activity / mitochondrial membrane organization / negative regulation of chaperone-mediated autophagy / regulation of reactive oxygen species biosynthetic process / positive regulation of protein localization to cell periphery / regulation of synaptic vesicle recycling / negative regulation of platelet-derived growth factor receptor signaling pathway / negative regulation of exocytosis / regulation of glutamate secretion / response to iron(II) ion / regulation of norepinephrine uptake / SNARE complex assembly / positive regulation of neurotransmitter secretion / dopamine biosynthetic process / regulation of locomotion / positive regulation of inositol phosphate biosynthetic process / synaptic vesicle priming / regulation of macrophage activation / negative regulation of microtubule polymerization / synaptic vesicle transport / dopamine uptake involved in synaptic transmission / dynein complex binding / regulation of dopamine secretion / positive regulation of receptor recycling / protein kinase inhibitor activity / negative regulation of thrombin-activated receptor signaling pathway / response to type II interferon / cuprous ion binding / positive regulation of exocytosis / synaptic vesicle exocytosis / positive regulation of endocytosis / kinesin binding / cysteine-type endopeptidase inhibitor activity involved in apoptotic process / response to magnesium ion / synaptic vesicle endocytosis / regulation of presynapse assembly / negative regulation of serotonin uptake / alpha-tubulin binding / phospholipid metabolic process / supramolecular fiber organization / axon terminus / mitochondrial ATP synthesis coupled electron transport / inclusion body / fatty acid metabolic process / cellular response to copper ion / cellular response to epinephrine stimulus / Hsp70 protein binding / response to interleukin-1 / adult locomotory behavior / negative regulation of cysteine-type endopeptidase activity involved in apoptotic process / positive regulation of release of sequestered calcium ion into cytosol / SNARE binding / excitatory postsynaptic potential / long-term synaptic potentiation / phosphoprotein binding / protein tetramerization / regulation of transmembrane transporter activity / synapse organization / microglial cell activation / negative regulation of protein kinase activity / regulation of long-term neuronal synaptic plasticity / protein destabilization / ferrous iron binding / tau protein binding / positive regulation of protein serine/threonine kinase activity / PKR-mediated signaling / receptor internalization / phospholipid binding / activation of cysteine-type endopeptidase activity involved in apoptotic process / synaptic vesicle membrane / positive regulation of inflammatory response / positive regulation of peptidyl-serine phosphorylation / actin cytoskeleton / actin binding / cellular response to oxidative stress / histone binding / cell cortex / growth cone / chemical synaptic transmission / neuron apoptotic process / negative regulation of neuron apoptotic process / transcription cis-regulatory region binding / postsynapse / amyloid fibril formation / response to lipopolysaccharide / molecular adaptor activity / oxidoreductase activity / lysosome / response to xenobiotic stimulus 類似検索 - 分子機能 | |||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||
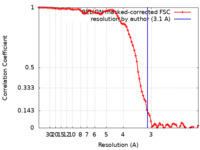
| 手法 | らせん対称体再構成法 / クライオ電子顕微鏡法 / 解像度: 3.1 Å | |||||||||
 データ登録者 データ登録者 | Pesch V / Reithofer S / Ma L / Flores-Fernandez JM / Oezduezenciler P / Busch Y / Lien Y / Rudtke O / Frieg B / Schroeder GF ...Pesch V / Reithofer S / Ma L / Flores-Fernandez JM / Oezduezenciler P / Busch Y / Lien Y / Rudtke O / Frieg B / Schroeder GF / Wille H / Tamgueney G | |||||||||
| 資金援助 |  米国, 米国,  ドイツ, 2件 ドイツ, 2件
| |||||||||
 引用 引用 |  ジャーナル: Brain / 年: 2024 ジャーナル: Brain / 年: 2024タイトル: Vaccination with structurally adapted fungal protein fibrils induces immunity to Parkinson's disease. 著者: Verena Pesch / José Miguel Flores-Fernandez / Sara Reithofer / Liang Ma / Pelin Özdüzenciler / Yannick Busch / Aishwarya Sriraman / YongLiang Wang / Sara Amidian / Chiara V M Kroepel / ...著者: Verena Pesch / José Miguel Flores-Fernandez / Sara Reithofer / Liang Ma / Pelin Özdüzenciler / Yannick Busch / Aishwarya Sriraman / YongLiang Wang / Sara Amidian / Chiara V M Kroepel / Laura Müller / Yi Lien / Olivia Rudtke / Benedikt Frieg / Gunnar F Schröder / Holger Wille / Gültekin Tamgüney /   要旨: The pathological misfolding and aggregation of soluble α-synuclein into toxic oligomers and insoluble amyloid fibrils causes Parkinson's disease, a progressive age-related neurodegenerative disease ...The pathological misfolding and aggregation of soluble α-synuclein into toxic oligomers and insoluble amyloid fibrils causes Parkinson's disease, a progressive age-related neurodegenerative disease for which there is no cure. HET-s is a soluble fungal protein that can form assembled amyloid fibrils in its prion state. We engineered HET-s(218-298) to form four different fibrillar vaccine candidates, each displaying a specific conformational epitope present on the surface of α-synuclein fibrils. Vaccination with these four vaccine candidates prolonged the survival of immunized TgM83+/- mice challenged with α-synuclein fibrils by 8% when injected into the brain to model brain-first Parkinson's disease or by 21% and 22% when injected into the peritoneum or gut wall, respectively, to model body-first Parkinson's disease. Antibodies from fully immunized mice recognized α-synuclein fibrils and brain homogenates from patients with Parkinson's disease, dementia with Lewy bodies and multiple system atrophy. Conformation-specific vaccines that mimic epitopes present only on the surface of pathological fibrils but not on soluble monomers, hold great promise for protection against Parkinson's disease, related synucleinopathies and other amyloidogenic protein misfolding disorders. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_17111.map.gz emd_17111.map.gz | 8.5 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-17111-v30.xml emd-17111-v30.xml emd-17111.xml emd-17111.xml | 14.8 KB 14.8 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_17111_fsc.xml emd_17111_fsc.xml | 8.9 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_17111.png emd_17111.png | 48.5 KB | ||
| Filedesc metadata |  emd-17111.cif.gz emd-17111.cif.gz | 5.3 KB | ||
| その他 |  emd_17111_half_map_1.map.gz emd_17111_half_map_1.map.gz emd_17111_half_map_2.map.gz emd_17111_half_map_2.map.gz | 46.2 MB 46.2 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17111 http://ftp.pdbj.org/pub/emdb/structures/EMD-17111 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17111 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17111 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_17111_validation.pdf.gz emd_17111_validation.pdf.gz | 803.9 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_17111_full_validation.pdf.gz emd_17111_full_validation.pdf.gz | 803.4 KB | 表示 | |
| XML形式データ |  emd_17111_validation.xml.gz emd_17111_validation.xml.gz | 14.7 KB | 表示 | |
| CIF形式データ |  emd_17111_validation.cif.gz emd_17111_validation.cif.gz | 20.8 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17111 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17111 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17111 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17111 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  8oqiMC M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_17111.map.gz / 形式: CCP4 / 大きさ: 59.6 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_17111.map.gz / 形式: CCP4 / 大きさ: 59.6 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Cryo-EM structure of the wild-type alpha-synuclein fibril. | ||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.06 Å | ||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-ハーフマップ: Cryo-EM structure of the wild-type alpha-synuclein fibril (half...
| ファイル | emd_17111_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Cryo-EM structure of the wild-type alpha-synuclein fibril (half map 2). | ||||||||||||
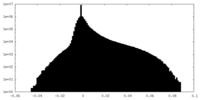
| 投影像・断面図 |
| ||||||||||||
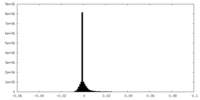
| 密度ヒストグラム |
-ハーフマップ: Cryo-EM structure of the wild-type alpha-synuclein fibril (half...
| ファイル | emd_17111_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Cryo-EM structure of the wild-type alpha-synuclein fibril (half map 1). | ||||||||||||
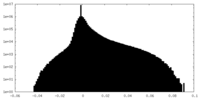
| 投影像・断面図 |
| ||||||||||||
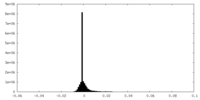
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Human wild-type alpha-synuclein fibril
| 全体 | 名称: Human wild-type alpha-synuclein fibril |
|---|---|
| 要素 |
|
-超分子 #1: Human wild-type alpha-synuclein fibril
| 超分子 | 名称: Human wild-type alpha-synuclein fibril / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: all 詳細: N-terminally acetylated human wild-type alpha-synuclein |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
-分子 #1: Alpha-synuclein
| 分子 | 名称: Alpha-synuclein / タイプ: protein_or_peptide / ID: 1 詳細: N-terminally acetylated human wild-type alpha-synuclein. コピー数: 10 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 14.476108 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MDVFMKGLSK AKEGVVAAAE KTKQGVAEAA GKTKEGVLYV GSKTKEGVVH GVATVAEKTK EQVTNVGGAV VTGVTAVAQK TVEGAGSIA AATGFVKKDQ LGKNEEGAPQ EGILEDMPVD PDNEAYEMPS EEGYQDYEPE A UniProtKB: Alpha-synuclein |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | らせん対称体再構成法 |
| 試料の集合状態 | filament |
- 試料調製
試料調製
| 緩衝液 | pH: 7.2 |
|---|---|
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K3 BIOQUANTUM (6k x 4k) 平均電子線量: 30.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 2.5 µm / 最小 デフォーカス(公称値): 0.5 µm |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー




 Z
Z Y
Y X
X