+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-13046 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of Brr2 in complex with Jab1/MPN and C9ORF78 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | mRNA Splicing / Spliceosomal Assembly / Splicing Regulation / Brr2 Helicase / SPLICING | |||||||||
| Function / homology |  Function and homology information Function and homology informationregulation of homologous chromosome segregation / cis assembly of pre-catalytic spliceosome / spliceosome conformational change to release U4 (or U4atac) and U1 (or U11) / U2-type catalytic step 1 spliceosome / RNA splicing, via transesterification reactions / mRNA cis splicing, via spliceosome / U2-type precatalytic spliceosome / U2-type catalytic step 2 spliceosome / K63-linked polyubiquitin modification-dependent protein binding / mRNA Splicing - Minor Pathway ...regulation of homologous chromosome segregation / cis assembly of pre-catalytic spliceosome / spliceosome conformational change to release U4 (or U4atac) and U1 (or U11) / U2-type catalytic step 1 spliceosome / RNA splicing, via transesterification reactions / mRNA cis splicing, via spliceosome / U2-type precatalytic spliceosome / U2-type catalytic step 2 spliceosome / K63-linked polyubiquitin modification-dependent protein binding / mRNA Splicing - Minor Pathway / chromosome, centromeric region / spliceosomal tri-snRNP complex assembly / U5 snRNA binding / U5 snRNP / U2 snRNA binding / U6 snRNA binding / pre-mRNA intronic binding / U1 snRNA binding / U4/U6 x U5 tri-snRNP complex / catalytic step 2 spliceosome / mRNA Splicing - Major Pathway / RNA splicing / helicase activity / chromosome segregation / spliceosomal complex / mRNA processing / mRNA splicing, via spliceosome / osteoblast differentiation / cellular response to tumor necrosis factor / cellular response to lipopolysaccharide / RNA helicase activity / RNA helicase / nuclear speck / ATP hydrolysis activity / RNA binding / nucleoplasm / ATP binding / identical protein binding / membrane / nucleus / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
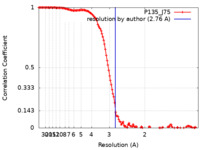
| Method | single particle reconstruction / cryo EM / Resolution: 2.76 Å | |||||||||
 Authors Authors | Bergfort A / Hilal T | |||||||||
| Funding support |  Germany, 1 items Germany, 1 items
| |||||||||
 Citation Citation |  Journal: Nucleic Acids Res / Year: 2022 Journal: Nucleic Acids Res / Year: 2022Title: The intrinsically disordered TSSC4 protein acts as a helicase inhibitor, placeholder and multi-interaction coordinator during snRNP assembly and recycling. Authors: Alexandra Bergfort / Tarek Hilal / Benno Kuropka / İbrahim Avşar Ilik / Gert Weber / Tuğçe Aktaş / Christian Freund / Markus C Wahl /  Abstract: Biogenesis of spliceosomal small nuclear ribonucleoproteins (snRNPs) and their recycling after splicing require numerous assembly/recycling factors whose modes of action are often poorly understood. ...Biogenesis of spliceosomal small nuclear ribonucleoproteins (snRNPs) and their recycling after splicing require numerous assembly/recycling factors whose modes of action are often poorly understood. The intrinsically disordered TSSC4 protein has been identified as a nuclear-localized U5 snRNP and U4/U6-U5 tri-snRNP assembly/recycling factor, but how TSSC4's intrinsic disorder supports TSSC4 functions remains unknown. Using diverse interaction assays and cryogenic electron microscopy-based structural analysis, we show that TSSC4 employs four conserved, non-contiguous regions to bind the PRPF8 Jab1/MPN domain and the SNRNP200 helicase at functionally important sites. It thereby inhibits SNRNP200 helicase activity, spatially aligns the proteins, coordinates formation of a U5 sub-module and transiently blocks premature interaction of SNRNP200 with at least three other spliceosomal factors. Guided by the structure, we designed a TSSC4 variant that lacks stable binding to the PRPF8 Jab1/MPN domain or SNRNP200 in vitro. Comparative immunoprecipitation/mass spectrometry from HEK293 nuclear extract revealed distinct interaction profiles of wild type TSSC4 and the variant deficient in PRPF8/SNRNP200 binding with snRNP proteins, other spliceosomal proteins as well as snRNP assembly/recycling factors and chaperones. Our findings elucidate molecular strategies employed by an intrinsically disordered protein to promote snRNP assembly, and suggest multiple TSSC4-dependent stages during snRNP assembly/recycling. #1:  Journal: Acta Crystallogr D Struct Biol / Year: 2019 Journal: Acta Crystallogr D Struct Biol / Year: 2019Title: Macromolecular structure determination using X-rays, neutrons and electrons: recent developments in Phenix. Authors: Liebschner D / Afonine PV / Baker ML / Bunkoczi G / Chen VB / Croll TI / Hintze B / Hung LW / Jain S / McCoy AJ / Moriarty NW / Oeffner RD / Poon BK / Prisant MG / Read RJ / Richardson JS / ...Authors: Liebschner D / Afonine PV / Baker ML / Bunkoczi G / Chen VB / Croll TI / Hintze B / Hung LW / Jain S / McCoy AJ / Moriarty NW / Oeffner RD / Poon BK / Prisant MG / Read RJ / Richardson JS / Richardson DC / Sammito MD / Sobolev OV / Stockwell DH / Terwilliger TC / Urzhumtsev AG / Videau LL / Williams CJ / Adams PD | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_13046.map.gz emd_13046.map.gz | 41.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-13046-v30.xml emd-13046-v30.xml emd-13046.xml emd-13046.xml | 21.5 KB 21.5 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_13046_fsc.xml emd_13046_fsc.xml | 13.3 KB | Display |  FSC data file FSC data file |
| Images |  emd_13046.png emd_13046.png | 101.7 KB | ||
| Filedesc metadata |  emd-13046.cif.gz emd-13046.cif.gz | 8.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-13046 http://ftp.pdbj.org/pub/emdb/structures/EMD-13046 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13046 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13046 | HTTPS FTP |
-Validation report
| Summary document |  emd_13046_validation.pdf.gz emd_13046_validation.pdf.gz | 467.7 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_13046_full_validation.pdf.gz emd_13046_full_validation.pdf.gz | 467.3 KB | Display | |
| Data in XML |  emd_13046_validation.xml.gz emd_13046_validation.xml.gz | 13.5 KB | Display | |
| Data in CIF |  emd_13046_validation.cif.gz emd_13046_validation.cif.gz | 18.1 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13046 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13046 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13046 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13046 | HTTPS FTP |
-Related structure data
| Related structure data |  7os2MC  7os1C  7px3C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_13046.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_13046.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 0.657 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Trimeric complex of Brr2, the Jab1/MPN domain of Prp8 and a N-ter...
| Entire | Name: Trimeric complex of Brr2, the Jab1/MPN domain of Prp8 and a N-terminal fragment of C9ORF78 |
|---|---|
| Components |
|
-Supramolecule #1: Trimeric complex of Brr2, the Jab1/MPN domain of Prp8 and a N-ter...
| Supramolecule | Name: Trimeric complex of Brr2, the Jab1/MPN domain of Prp8 and a N-terminal fragment of C9ORF78 type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #2: U5 small nuclear ribonucleoprotein 200 kDa helicase (Brr2)
| Supramolecule | Name: U5 small nuclear ribonucleoprotein 200 kDa helicase (Brr2) type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #3: Telomere length and silencing protein 1 homolog (C9ORF78)
| Supramolecule | Name: Telomere length and silencing protein 1 homolog (C9ORF78) type: complex / ID: 3 / Parent: 1 / Macromolecule list: #2 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #4: Pre-mRNA-processing-splicing factor 8 (Prp8) Jab1/MPN Domain
| Supramolecule | Name: Pre-mRNA-processing-splicing factor 8 (Prp8) Jab1/MPN Domain type: complex / ID: 4 / Parent: 1 / Macromolecule list: #3 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: U5 small nuclear ribonucleoprotein 200 kDa helicase
| Macromolecule | Name: U5 small nuclear ribonucleoprotein 200 kDa helicase / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO / EC number: RNA helicase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 198.785797 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GAEFDLDQGG EALAPRQVLD LEDLVFTQGS HFMANKRCQL PDGSFRRQRK GYEEVHVPAL KPKPFGSEEQ LLPVEKLPKY AQAGFEGFK TLNRIQSKLY RAALETDENL LLCAPTGAGK TNVALMCMLR EIGKHINMDG TINVDDFKII YIAPMRSLVQ E MVGSFGKR ...String: GAEFDLDQGG EALAPRQVLD LEDLVFTQGS HFMANKRCQL PDGSFRRQRK GYEEVHVPAL KPKPFGSEEQ LLPVEKLPKY AQAGFEGFK TLNRIQSKLY RAALETDENL LLCAPTGAGK TNVALMCMLR EIGKHINMDG TINVDDFKII YIAPMRSLVQ E MVGSFGKR LATYGITVAE LTGDHQLCKE EISATQIIVC TPEKWDIITR KGGERTYTQL VRLIILDEIH LLHDDRGPVL EA LVARAIR NIEMTQEDVR LIGLSATLPN YEDVATFLRV DPAKGLFYFD NSFRPVPLEQ TYVGITEKKA IKRFQIMNEI VYE KIMEHA GKNQVLVFVH SRKETGKTAR AIRDMCLEKD TLGLFLREGS ASTEVLRTEA EQCKNLELKD LLPYGFAIHH AGMT RVDRT LVEDLFADKH IQVLVSTATL AWGVNLPAHT VIIKGTQVYS PEKGRWTELG ALDILQMLGR AGRPQYDTKG EGILI TSHG ELQYYLSLLN QQLPIESQMV SKLPDMLNAE IVLGNVQNAK DAVNWLGYAY LYIRMLRSPT LYGISHDDLK GDPLLD QRR LDLVHTAALM LDKNNLVKYD KKTGNFQVTE LGRIASHYYI TNDTVQTYNQ LLKPTLSEIE LFRVFSLSSE FKNITVR EE EKLELQKLLE RVPIPVKESI EEPSAKINVL LQAFISQLKL EGFALMADMV YVTQSAGRLM RAIFEIVLNR GWAQLTDK T LNLCKMIDKR MWQSMCPLRQ FRKLPEEVVK KIEKKNFPFE RLYDLNHNEI GELIRMPKMG KTIHKYVHLF PKLELSVHL QPITRSTLKV ELTITPDFQW DEKVHGSSEA FWILVEDVDS EVILHHEYFL LKAKYAQDEH LITFFVPVFE PLPPQYFIRV VSDRWLSCE TQLPVSFRHL ILPEKYPPPT ELLDLQPLPV SALRNSAFES LYQDKFPFFN PIQTQVFNTV YNSDDNVFVG A PTGSGKTI CAEFAILRML LQSSEGRCVY ITPMEALAEQ VYMDWYEKFQ DRLNKKVVLL TGETSTDLKL LGKGNIIIST PE KWDILSR RWKQRKNVQN INLFVVDEVH LIGGENGPVL EVICSRMRYI SSQIERPIRI VALSSSLSNA KDVAHWLGCS ATS TFNFHP NVRPVPLELH IQGFNISHTQ TRLLSMAKPV YHAITKHSPK KPVIVFVPSR KQTRLTAIDI LTTCAADIQR QRFL HCTEK DLIPYLEKLS DSTLKETLLN GVGYLHEGLS PMERRLVEQL FSSGAIQVVV ASRSLCWGMN VAAHLVIIMD TQYYN GKIH AYVDYPIYDV LQMVGHANRP LQDDEGRCVI MCQGSKKDFF KKFLYEPLPV ESHLDHCMHD HFNAEIVTKT IENKQD AVD YLTWTFLYRR MTQNPNYYNL QGISHRHLSD HLSELVEQTL SDLEQSKCIS IEDEMDVAPL NLGMIAAYYY INYTTIE LF SMSLNAKTKV RGLIEIISNA AEYENIPIRH HEDNLLRQLA QKVPHKLNNP KFNDPHVKTN LLLQAHLSRM QLSAELQS D TEEILSKAIR LIQACVDVLS SNGWLSPALA AMELAQMVTQ AMWSKDSYLK QLPHFTSEHI KRCTDKGVES VFDIMEMED EERNALLQLT DSQIADVARF CNRYPNIELS YEVVDKDSIR SGGPVVVLVQ LEREEEVTGP VIAPLFPQKR EEGWWVVIGD AKSNSLISI KRLTLQQKAK VKLDFVAPAT GAHNYTLYFM SDAYMGCDQE YKFSVDVKEA UniProtKB: U5 small nuclear ribonucleoprotein 200 kDa helicase |
-Macromolecule #2: Telomere length and silencing protein 1 homolog
| Macromolecule | Name: Telomere length and silencing protein 1 homolog / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 34.081516 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GAMAMPVVRK IFRRRRGDSE SEEDEQDSEE VRLKLEETRE VQNLRKRPNG VSAVALLVGE KVQEETTLVD DPFQMKTGGM VDMKKLKER GKDKISEEED LHLGTSFSAE TNRRDEDADM MKYIETELKK RKGIVEHEEQ KVKPKNAEDC LYELPENIRV S SAKKTEEM ...String: GAMAMPVVRK IFRRRRGDSE SEEDEQDSEE VRLKLEETRE VQNLRKRPNG VSAVALLVGE KVQEETTLVD DPFQMKTGGM VDMKKLKER GKDKISEEED LHLGTSFSAE TNRRDEDADM MKYIETELKK RKGIVEHEEQ KVKPKNAEDC LYELPENIRV S SAKKTEEM LSNQMLSGIP EVDLGIDAKI KNIISTEDAK ARLLAEQQNK KKDSETSFVP TNMAVNYVQH NRFYHEELNA PI RRNKEEP KARPLRVGDT EKPEPERSPP NRKRPANEKA TDDYHYEKFK KMNRRY UniProtKB: Splicing factor C9orf78 |
-Macromolecule #3: Pre-mRNA-processing-splicing factor 8
| Macromolecule | Name: Pre-mRNA-processing-splicing factor 8 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 31.860984 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GPLGSMTQTF SSKTEWRVRA ISAANLHLRT NHIYVSSDDI KETGYTYILP KNVLKKFICI SDLRAQIAGY LYGVSPPDNP QVKEIRCIV MVPQWGTHQT VHLPGQLPQH EYLKEMEPLG WIHTQPNESP QLSPQDVTTH AKIMADNPSW DGEKTIIITC S FTPGSCTL ...String: GPLGSMTQTF SSKTEWRVRA ISAANLHLRT NHIYVSSDDI KETGYTYILP KNVLKKFICI SDLRAQIAGY LYGVSPPDNP QVKEIRCIV MVPQWGTHQT VHLPGQLPQH EYLKEMEPLG WIHTQPNESP QLSPQDVTTH AKIMADNPSW DGEKTIIITC S FTPGSCTL TAYKLTPSGY EWGRQNTDKG NNPKGYLPSH YERVQMLLSD RFLGFFMVPA QSSWNYNFMG VRHDPNMKYE LQ LANPKEF YHEVHRPSHF LNFALLQEGE VYSADREDLY A UniProtKB: Pre-mRNA-processing-splicing factor 8 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 5 mg/mL |
|---|---|
| Buffer | pH: 7.6 |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 283 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: COUNTING / Number grids imaged: 1 / Number real images: 5160 / Average exposure time: 40.0 sec. / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.8 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 120000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model |
| ||||||
|---|---|---|---|---|---|---|---|
| Refinement | Space: REAL / Protocol: OTHER / Overall B value: 102 / Target criteria: CC, R.m.s.d. Bonds / Angles | ||||||
| Output model |  PDB-7os2: |
 Movie
Movie Controller
Controller