+Search query
-Structure paper
| Title | Structure of the antenna complex expressed during far-red light photoacclimation in Synechococcus sp. PCC 7335. |
|---|---|
| Journal, issue, pages | J Biol Chem, Vol. 300, Issue 2, Page 105590, Year 2024 |
| Publish date | Dec 22, 2023 |
 Authors Authors | Christopher J Gisriel / Gaozhong Shen / Gary W Brudvig / Donald A Bryant /  |
| PubMed Abstract | Far-red light photoacclimation, or FaRLiP, is a facultative response exhibited by some cyanobacteria that allows them to absorb and utilize lower energy light (700-800 nm) than the wavelengths ...Far-red light photoacclimation, or FaRLiP, is a facultative response exhibited by some cyanobacteria that allows them to absorb and utilize lower energy light (700-800 nm) than the wavelengths typically used for oxygenic photosynthesis (400-700 nm). During this process, three essential components of the photosynthetic apparatus are altered: photosystem I, photosystem II, and the phycobilisome. In all three cases, at least some of the chromophores found in these pigment-protein complexes are replaced by chromophores that have red-shifted absorbance relative to the analogous complexes produced in visible light. Recent structural and spectroscopic studies have elucidated important features of the two photosystems when altered to absorb and utilize far-red light, but much less is understood about the modified phycobiliproteins made during FaRLiP. We used single-particle, cryo-EM to determine the molecular structure of a phycobiliprotein core complex comprising allophycocyanin variants that absorb far-red light during FaRLiP in the marine cyanobacterium Synechococcus sp. PCC 7335. The structure reveals the arrangement of the numerous red-shifted allophycocyanin variants and the probable locations of the chromophores that serve as the terminal emitters in this complex. It also suggests how energy is transferred to the photosystem II complexes produced during FaRLiP. The structure additionally allows comparisons with other previously studied allophycocyanins to gain insights into how phycocyanobilin chromophores can be tuned to absorb far-red light. These studies provide new insights into how far-red light is harvested and utilized during FaRLiP, a widespread cyanobacterial photoacclimation mechanism. |
 External links External links |  J Biol Chem / J Biol Chem /  PubMed:38141759 / PubMed:38141759 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 2.35 - 2.78 Å |
| Structure data | EMDB-42278, PDB-8uhe: EMDB-42281, PDB-8uhi: |
| Chemicals | 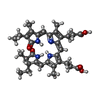 ChemComp-CYC: 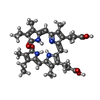 ChemComp-M1V:  ChemComp-CL: 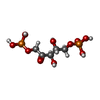 ChemComp-RUB:  ChemComp-MG:  ChemComp-HOH: |
| Source |
|
 Keywords Keywords | PHOTOSYNTHESIS / light-harvesting / allophycocyanin / phycocyanobilin |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers







 synechococcus sp. pcc 7335 (bacteria)
synechococcus sp. pcc 7335 (bacteria)