+Search query
-Structure paper
| Title | Structural and biochemical analysis of Bacillus anthracis prephenate dehydrogenase reveals an unusual mode of inhibition by tyrosine via the ACT domain. |
|---|---|
| Journal, issue, pages | Febs J., Vol. 287, Page 2235-2255, Year 2020 |
| Publish date | Feb 24, 2017 (structure data deposition date) |
 Authors Authors | Shabalin, I.G. / Gritsunov, A. / Hou, J. / Slawek, J. / Miks, C.D. / Cooper, D.R. / Minor, W. / Christendat, D. |
 External links External links |  Febs J. / Febs J. /  PubMed:31750992 PubMed:31750992 |
| Methods | X-ray diffraction |
| Resolution | 2.01 - 2.75 Å |
| Structure data |  PDB-5uyy:  PDB-5v0s:  PDB-6cxd:  PDB-6u60: |
| Chemicals | 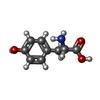 ChemComp-TYR:  ChemComp-HOH:  ChemComp-SO4:  ChemComp-CA:  ChemComp-PO4:  ChemComp-NAD: |
| Source |
|
 Keywords Keywords | OXIDOREDUCTASE / prephenate dehydrogenase / tyrA / Bacillus anthracis / CSGID / Structural Genomics / Center for Structural Genomics of Infectious Diseases / HYDROLASE / peptidase B |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers




