+Search query
-Structure paper
| Title | Crystal structure of the thioesterification conformation of Bacillus subtilis o-succinylbenzoyl-CoA synthetase reveals a distinct substrate-binding mode |
|---|---|
| Journal, issue, pages | J. Biol. Chem., Vol. 292, Page 12296-12310, Year 2017 |
| Publish date | Mar 2, 2017 (structure data deposition date) |
 Authors Authors | Chen, Y. / Li, T.L. / Lin, X. / Li, X. / Li, X.D. / Guo, Z. |
 External links External links |  J. Biol. Chem. / J. Biol. Chem. /  PubMed:28559280 PubMed:28559280 |
| Methods | X-ray diffraction |
| Resolution | 1.763 - 1.9 Å |
| Structure data |  PDB-5x8f:  PDB-5x8g: |
| Chemicals | 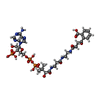 ChemComp-S0N:  ChemComp-AMP:  ChemComp-MG:  ChemComp-NA:  ChemComp-PEG:  ChemComp-CA:  ChemComp-HOH: |
| Source |
|
 Keywords Keywords | LIGASE / Adenylate-forming Enzyme / acyl-CoA synthetase / thioester conformation / CoA / pantetheine tunnel / ADP binding subsite / domain alternation / large conformational change / inter-domain linker |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers




