+Search query
-Structure paper
| Title | An interfacial-intramolecular electron highway for accelerated electrocatalytic CO reduction by an O-tolerant formate dehydrogenase. |
|---|---|
| Journal, issue, pages | Nat Commun, Vol. 17, Issue 1, Year 2026 |
| Publish date | Mar 3, 2026 |
 Authors Authors | Weisong Liu / Peng Zhang / Xiufeng Wang / Kuncheng Zhang / Wenhua Yang / Huijuan Cui / Jun Liu / Junsong Sun / Chun You / Haiyang Cui / Zhiguang Zhu / Lingling Zhang /  |
| PubMed Abstract | Bioelectrocatalytic CO reduction offers a sustainable route for CO bioconversion, yet remains limited by interfacial-intramolecular electron transfer and oxygen sensitivity. Here, we mine a formate ...Bioelectrocatalytic CO reduction offers a sustainable route for CO bioconversion, yet remains limited by interfacial-intramolecular electron transfer and oxygen sensitivity. Here, we mine a formate dehydrogenase from Shewanella oneidensis MR-1 (SoFdhAB) featuring completely oxygen tolerant and direct-electron-transfer (DET) electrocatalytic performances. Cryo-electron microscopy (Cryo-EM) analysis reveals an intramolecular electron highway comprising five [4Fe-4S] clusters, a regional face-face contact facilitating interfacial ET, and a unique oxygen resistance mechanism different from inactivation-activation. By acquiring a beneficial variant SoFdhAB-Y94S, a direct bioelectrocatalytic CO reduction system is constructed, accumulating 2.88 ± 0.03 mmol formate in 64 hours with a steady rate of 45.3 ± 0.5 μmol h cm and a Faradaic efficiency of 93.1 ± 5.2%. The merits of oxygen tolerance and efficient (electro)catalytic property endow SoFdhAB a robust enzyme adopted in potential application scenarios, and the inherent DET capability may inspire the interfacial engineering of other oxidoreductases. |
 External links External links |  Nat Commun / Nat Commun /  PubMed:41775696 / PubMed:41775696 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 2.75 Å |
| Structure data | EMDB-64903, PDB-9vap: |
| Chemicals | 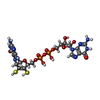 ChemComp-MGD:  ChemComp-SF4: 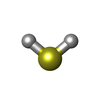 ChemComp-H2S:  ChemComp-W: |
| Source |
|
 Keywords Keywords | OXIDOREDUCTASE / Formate dehydrogenase / carbon dioxide reduction / tungsten (W)-dependent |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers





 shewanella oneidensis mr-1 (bacteria)
shewanella oneidensis mr-1 (bacteria)