+Search query
-Structure paper
| Title | Structural basis for the conformational protection of nitrogenase from O. |
|---|---|
| Journal, issue, pages | Nature, Vol. 637, Issue 8047, Page 991-997, Year 2025 |
| Publish date | Jan 8, 2025 |
 Authors Authors | Sarah M Narehood / Brian D Cook / Suppachai Srisantitham / Vanessa H Eng / Angela A Shiau / Kelly L McGuire / R David Britt / Mark A Herzik / F Akif Tezcan /  |
| PubMed Abstract | The low reduction potentials required for the reduction of dinitrogen (N) render metal-based nitrogen-fixation catalysts vulnerable to irreversible damage by dioxygen (O). Such O sensitivity ...The low reduction potentials required for the reduction of dinitrogen (N) render metal-based nitrogen-fixation catalysts vulnerable to irreversible damage by dioxygen (O). Such O sensitivity represents a major conundrum for the enzyme nitrogenase, as a large fraction of nitrogen-fixing organisms are either obligate aerobes or closely associated with O-respiring organisms to support the high energy demand of catalytic N reduction. To counter O damage to nitrogenase, diazotrophs use O scavengers, exploit compartmentalization or maintain high respiration rates to minimize intracellular O concentrations. A last line of damage control is provided by the 'conformational protection' mechanism, in which a [2Fe:2S] ferredoxin-family protein termed FeSII (ref. ) is activated under O stress to form an O-resistant complex with the nitrogenase component proteins. Despite previous insights, the molecular basis for the conformational O protection of nitrogenase and the mechanism of FeSII activation are not understood. Here we report the structural characterization of the Azotobacter vinelandii FeSII-nitrogenase complex by cryo-electron microscopy. Our studies reveal a core complex consisting of two molybdenum-iron proteins (MoFePs), two iron proteins (FePs) and one FeSII homodimer, which polymerize into extended filaments. In this three-protein complex, FeSII mediates an extensive network of interactions with MoFeP and FeP to position their iron-sulphur clusters in catalytically inactive but O-protected states. The architecture of the FeSII-nitrogenase complex is confirmed by solution studies, which further indicate that the activation of FeSII involves an oxidation-induced conformational change. |
 External links External links |  Nature / Nature /  PubMed:39779844 / PubMed:39779844 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 2.27 - 3.94 Å |
| Structure data | EMDB-45923, PDB-9ctz: EMDB-45924, PDB-9cu0: EMDB-45925, PDB-9cu1: EMDB-45926, PDB-9cu2: |
| Chemicals | 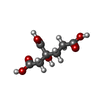 ChemComp-HCA: 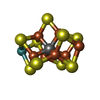 ChemComp-ICS:  ChemComp-FE: 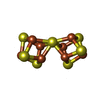 ChemComp-CLF:  ChemComp-HOH:  ChemComp-ADP:  ChemComp-MG:  ChemComp-SF4:  ChemComp-FES: |
| Source |
|
 Keywords Keywords | METAL BINDING PROTEIN / Nitrogenase / FeMoCo / nitrogen / P-cluster |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers











 azotobacter vinelandii (bacteria)
azotobacter vinelandii (bacteria)