+Search query
-Structure paper
| Title | Comparative structural analysis of human Na1.1 and Na1.5 reveals mutational hotspots for sodium channelopathies. |
|---|---|
| Journal, issue, pages | Proc Natl Acad Sci U S A, Vol. 118, Issue 11, Year 2021 |
| Publish date | Mar 16, 2021 |
 Authors Authors | Xiaojing Pan / Zhangqiang Li / Xueqin Jin / Yanyu Zhao / Gaoxingyu Huang / Xiaoshuang Huang / Zilin Shen / Yong Cao / Mengqiu Dong / Jianlin Lei / Nieng Yan /   |
| PubMed Abstract | Among the nine subtypes of human voltage-gated sodium (Na) channels, the brain and cardiac isoforms, Na1.1 and Na1.5, each carry more than 400 missense mutations respectively associated with epilepsy ...Among the nine subtypes of human voltage-gated sodium (Na) channels, the brain and cardiac isoforms, Na1.1 and Na1.5, each carry more than 400 missense mutations respectively associated with epilepsy and cardiac disorders. High-resolution structures are required for structure-function relationship dissection of the disease variants. We report the cryo-EM structures of the full-length human Na1.1-β4 complex at 3.3 Å resolution here and the Na1.5-E1784K variant in the accompanying paper. Up to 341 and 261 disease-related missense mutations in Na1.1 and Na1.5, respectively, are resolved. Comparative structural analysis reveals several clusters of disease mutations that are common to both Na1.1 and Na1.5. Among these, the majority of mutations on the extracellular loops above the pore domain and the supporting segments for the selectivity filter may impair structural integrity, while those on the pore domain and the voltage-sensing domains mostly interfere with electromechanical coupling and fast inactivation. Our systematic structural delineation of these mutations provides important insight into their pathogenic mechanism, which will facilitate the development of precise therapeutic interventions against various sodium channelopathies. |
 External links External links |  Proc Natl Acad Sci U S A / Proc Natl Acad Sci U S A /  PubMed:33712547 / PubMed:33712547 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 3.3 Å |
| Structure data | EMDB-30851, PDB-7dtd: |
| Chemicals |  ChemComp-NAG: 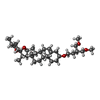 ChemComp-9Z9: |
| Source |
|
 Keywords Keywords | MEMBRANE PROTEIN / Voltage-gated sodium channel |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers





 homo sapiens (human)
homo sapiens (human)