+Search query
-Structure paper
| Title | Cryo-EM structures of lipopolysaccharide transporter LptBFGC in lipopolysaccharide or AMP-PNP-bound states reveal its transport mechanism. |
|---|---|
| Journal, issue, pages | Nat Commun, Vol. 10, Issue 1, Page 4175, Year 2019 |
| Publish date | Sep 13, 2019 |
 Authors Authors | Xiaodi Tang / Shenghai Chang / Qinghua Luo / Zhengyu Zhang / Wen Qiao / Caihuang Xu / Changbin Zhang / Yang Niu / Wenxian Yang / Ting Wang / Zhibo Zhang / Xiaofeng Zhu / Xiawei Wei / Changjiang Dong / Xing Zhang / Haohao Dong /   |
| PubMed Abstract | Lipopolysaccharides (LPS) of Gram-negative bacteria are critical for the defence against cytotoxic substances and must be transported from the inner membrane (IM) to the outer membrane (OM) through ...Lipopolysaccharides (LPS) of Gram-negative bacteria are critical for the defence against cytotoxic substances and must be transported from the inner membrane (IM) to the outer membrane (OM) through a bridge formed by seven membrane proteins (LptBFGCADE). The IM component LptBFG powers the process through a yet unclarified mechanism. Here we report three high-resolution cryo-EM structures of LptBFG alone and complexed with LptC (LptBFGC), trapped in either the LPS- or AMP-PNP-bound state. The structures reveal conformational changes between these states and substrate binding with or without LptC. We identify two functional transmembrane arginine-containing loops interacting with the bound AMP-PNP and elucidate allosteric communications between the domains. AMP-PNP binding induces an inward rotation and shift of the transmembrane helices of LptFG and LptC to tighten the cavity, with the closure of two lateral gates, to eventually expel LPS into the bridge. Functional assays reveal the functionality of the LptF and LptG periplasmic domains. Our findings shed light on the LPS transport mechanism. |
 External links External links |  Nat Commun / Nat Commun /  PubMed:31519889 / PubMed:31519889 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 3.1 - 3.7 Å |
| Structure data | EMDB-10121, PDB-6s8g: EMDB-10122, PDB-6s8h: EMDB-10125, PDB-6s8n: |
| Chemicals |  ChemComp-ANP: 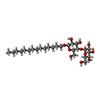 ChemComp-LMD:  ChemComp-LMN:  ChemComp-JSG: 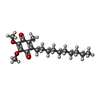 ChemComp-DCQ:  ChemComp-LMT: 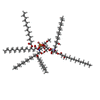 ChemComp-L0W: |
| Source |
|
 Keywords Keywords | TRANSPORT PROTEIN / lipopolysaccharide transporter / LPS / LptB2FGC / LptB / LptBFG / outer membrane / Gram-negative bacteria / ABC transporter / Inner membrane protein complex |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers









 shigella flexneri (bacteria)
shigella flexneri (bacteria)