+Search query
-Structure paper
| Title | Structural complexity of the co-chaperone SGTA: a conserved C-terminal region is implicated in dimerization and substrate quality control. |
|---|---|
| Journal, issue, pages | BMC Biol, Vol. 16, Issue 1, Page 76, Year 2018 |
| Publish date | Jul 11, 2018 |
 Authors Authors | Santiago Martínez-Lumbreras / Ewelina M Krysztofinska / Arjun Thapaliya / Alessandro Spilotros / Dijana Matak-Vinkovic / Enrico Salvadori / Peristera Roboti / Yvonne Nyathi / Janina H Muench / Maxie M Roessler / Dmitri I Svergun / Stephen High / Rivka L Isaacson /   |
| PubMed Abstract | BACKGROUND: Protein quality control mechanisms are essential for cell health and involve delivery of proteins to specific cellular compartments for recycling or degradation. In particular, stray ...BACKGROUND: Protein quality control mechanisms are essential for cell health and involve delivery of proteins to specific cellular compartments for recycling or degradation. In particular, stray hydrophobic proteins are captured in the aqueous cytosol by a co-chaperone, the small glutamine-rich, tetratricopeptide repeat-containing protein alpha (SGTA), which facilitates the correct targeting of tail-anchored membrane proteins, as well as the sorting of membrane and secretory proteins that mislocalize to the cytosol and endoplasmic reticulum-associated degradation. Full-length SGTA has an unusual elongated dimeric structure that has, until now, evaded detailed structural analysis. The C-terminal region of SGTA plays a key role in binding a broad range of hydrophobic substrates, yet in contrast to the well-characterized N-terminal and TPR domains, there is a lack of structural information on the C-terminal domain. In this study, we present new insights into the conformation and organization of distinct domains of SGTA and show that the C-terminal domain possesses a conserved region essential for substrate processing in vivo. RESULTS: We show that the C-terminal domain region is characterized by α-helical propensity and an intrinsic ability to dimerize independently of the N-terminal domain. Based on the properties of different regions of SGTA that are revealed using cell biology, NMR, SAXS, Native MS, and EPR, we observe that its C-terminal domain can dimerize in the full-length protein and propose that this reflects a closed conformation of the substrate-binding domain. CONCLUSION: Our results provide novel insights into the structural complexity of SGTA and provide a new basis for mechanistic studies of substrate binding and release at the C-terminal region. |
 External links External links |  BMC Biol / BMC Biol /  PubMed:29996828 / PubMed:29996828 /  PubMed Central PubMed Central |
| Methods | SAS (X-ray synchrotron) |
| Structure data | 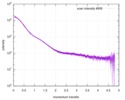 SASDDB6: Small glutamine-rich tetratricopeptide repeat-containing protein alpha (full length; SGTA_FL) 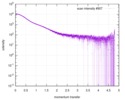 SASDDC6: Small glutamine-rich tetratricopeptide repeat-containing protein alpha (N-terminal-TPR domains; SGTA_NT) |
| Source |
|
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers



 Homo sapiens (human)
Homo sapiens (human)