+検索条件
-Structure paper
| タイトル | Cryo-EM structure of the human Asc-1 transporter complex. |
|---|---|
| ジャーナル・号・ページ | Nat Commun, Vol. 15, Issue 1, Page 3036, Year 2024 |
| 掲載日 | 2024年4月8日 |
 著者 著者 | Yaning Li / Yingying Guo / Angelika Bröer / Lu Dai / Stefan Brӧer / Renhong Yan /   |
| PubMed 要旨 | The Alanine-Serine-Cysteine transporter 1 (Asc-1 or SLC7A10) forms a crucial heterodimeric transporter complex with 4F2hc (SLC3A2) through a covalent disulfide bridge. This complex enables the sodium- ...The Alanine-Serine-Cysteine transporter 1 (Asc-1 or SLC7A10) forms a crucial heterodimeric transporter complex with 4F2hc (SLC3A2) through a covalent disulfide bridge. This complex enables the sodium-independent transport of small neutral amino acids, including L-Alanine (L-Ala), Glycine (Gly), and D-Serine (D-Ser), within the central nervous system (CNS). D-Ser and Gly are two key endogenous glutamate co-agonists that activate N-methyl-d-aspartate (NMDA) receptors by binding to the allosteric site. Mice deficient in Asc-1 display severe symptoms such as tremors, ataxia, and seizures, leading to early postnatal death. Despite its physiological importance, the functional mechanism of the Asc-1-4F2hc complex has remained elusive. Here, we present cryo-electron microscopy (cryo-EM) structures of the human Asc-1-4F2hc complex in its apo state, D-Ser bound state, and L-Ala bound state, resolved at 3.6 Å, 3.5 Å, and 3.4 Å, respectively. Through detailed structural analysis and transport assays, we uncover a comprehensive alternating access mechanism that underlies conformational changes in the complex. In summary, our findings reveal the architecture of the Asc-1 and 4F2hc complex and provide valuable insights into substrate recognition and the functional cycle of this essential transporter complex. |
 リンク リンク |  Nat Commun / Nat Commun /  PubMed:38589439 / PubMed:38589439 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.42 - 3.5 Å |
| 構造データ | EMDB-37671, PDB-8wns: EMDB-37672, PDB-8wnt: EMDB-37675, PDB-8wny: |
| 化合物 |  ChemComp-NAG: 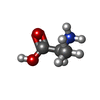 ChemComp-ALA: 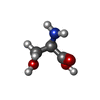 ChemComp-DSN: |
| 由来 |
|
 キーワード キーワード | MEMBRANE PROTEIN / SLC7A10 / SLC3A2 / ASC-1 |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について









 homo sapiens (ヒト)
homo sapiens (ヒト)