+検索条件
-Structure paper
| タイトル | A small-molecule activation mechanism that directly opens the KCNQ2 channel. |
|---|---|
| ジャーナル・号・ページ | Nat Chem Biol, Vol. 20, Issue 7, Page 847-856, Year 2024 |
| 掲載日 | 2024年1月2日 |
 著者 著者 | Shaoying Zhang / Demin Ma / Kun Wang / Ya Li / Zhenni Yang / Xiaoxiao Li / Junnan Li / Jiangnan He / Lianghe Mei / Yangliang Ye / Zongsheng Chen / Juwen Shen / Panpan Hou / Jiangtao Guo / Qiansen Zhang / Huaiyu Yang /  |
| PubMed 要旨 | Pharmacological activation of voltage-gated ion channels by ligands serves as the basis for therapy and mainly involves a classic gating mechanism that augments the native voltage-dependent open ...Pharmacological activation of voltage-gated ion channels by ligands serves as the basis for therapy and mainly involves a classic gating mechanism that augments the native voltage-dependent open probability. Through structure-based virtual screening, we identified a new scaffold compound, Ebio1, serving as a potent and subtype-selective activator for the voltage-gated potassium channel KCNQ2 and featuring a new activation mechanism. Single-channel patch-clamp, cryogenic-electron microscopy and molecular dynamic simulations, along with chemical derivatives, reveal that Ebio1 engages the KCNQ2 activation by generating an extended channel gate with a larger conductance at the saturating voltage (+50 mV). This mechanism is different from the previously observed activation mechanism of ligands on voltage-gated ion channels. Ebio1 caused S6 helices from residues S303 and F305 to perform a twist-to-open movement, which was sufficient to open the KCNQ2 gate. Overall, our findings provide mechanistic insights into the activation of KCNQ2 channel by Ebio1 and lend support for KCNQ-related drug development. |
 リンク リンク |  Nat Chem Biol / Nat Chem Biol /  PubMed:38167918 PubMed:38167918 |
| 手法 | EM (単粒子) |
| 解像度 | 3.0 - 3.4 Å |
| 構造データ | EMDB-35487, PDB-8ijk: EMDB-38041, PDB-8x43: |
| 化合物 |  ChemComp-7PN: 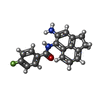 ChemComp-7Q0: |
| 由来 |
|
 キーワード キーワード | MEMBRANE PROTEIN / Potassium voltage-gated channel subfamily KQT member 2 / Ebio1 / Ebio1-S1 |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について







 homo sapiens (ヒト)
homo sapiens (ヒト)