+検索条件
-Structure paper
| タイトル | Conformational transitions and allosteric modulation in a heteromeric glycine receptor. |
|---|---|
| ジャーナル・号・ページ | Nat Commun, Vol. 14, Issue 1, Page 1363, Year 2023 |
| 掲載日 | 2023年3月13日 |
 著者 著者 | Eric Gibbs / Emily Klemm / David Seiferth / Arvind Kumar / Serban L Ilca / Philip C Biggin / Sudha Chakrapani /   |
| PubMed 要旨 | Glycine Receptors (GlyRs) provide inhibitory neuronal input in the spinal cord and brainstem, which is critical for muscle coordination and sensory perception. Synaptic GlyRs are a heteromeric ...Glycine Receptors (GlyRs) provide inhibitory neuronal input in the spinal cord and brainstem, which is critical for muscle coordination and sensory perception. Synaptic GlyRs are a heteromeric assembly of α and β subunits. Here we present cryo-EM structures of full-length zebrafish α1βGlyR in the presence of an antagonist (strychnine), agonist (glycine), or agonist with a positive allosteric modulator (glycine/ivermectin). Each structure shows a distinct pore conformation with varying degrees of asymmetry. Molecular dynamic simulations found the structures were in a closed (strychnine) and desensitized states (glycine and glycine/ivermectin). Ivermectin binds at all five interfaces, but in a distinct binding pose at the β-α interface. Subunit-specific features were sufficient to solve structures without a fiduciary marker and to confirm the 4α:1β stoichiometry recently observed. We also report features of the extracellular and intracellular domains. Together, our results show distinct compositional and conformational properties of αβGlyR and provide a framework for further study of this physiologically important channel. |
 リンク リンク |  Nat Commun / Nat Commun /  PubMed:36914669 / PubMed:36914669 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.0 Å |
| 構造データ | EMDB-29019, PDB-8fe1: |
| 化合物 |  ChemComp-GLY:  ChemComp-NAG: 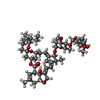 ChemComp-IVM: 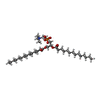 ChemComp-PX4:  ChemComp-PLM:  ChemComp-D10: |
| 由来 |
|
 キーワード キーワード | MEMBRANE PROTEIN / Glycine / Channel / Ivermectin / Pentameric |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について






