+検索条件
-Structure paper
| タイトル | TnpB structure reveals minimal functional core of Cas12 nuclease family. |
|---|---|
| ジャーナル・号・ページ | Nature, Vol. 616, Issue 7956, Page 384-389, Year 2023 |
| 掲載日 | 2023年4月5日 |
 著者 著者 | Giedrius Sasnauskas / Giedre Tamulaitiene / Gytis Druteika / Arturo Carabias / Arunas Silanskas / Darius Kazlauskas / Česlovas Venclovas / Guillermo Montoya / Tautvydas Karvelis / Virginijus Siksnys /  |
| PubMed 要旨 | The widespread TnpB proteins of IS200/IS605 transposon family have recently emerged as the smallest RNA-guided nucleases capable of targeted genome editing in eukaryotic cells. Bioinformatic analysis ...The widespread TnpB proteins of IS200/IS605 transposon family have recently emerged as the smallest RNA-guided nucleases capable of targeted genome editing in eukaryotic cells. Bioinformatic analysis identified TnpB proteins as the likely predecessors of Cas12 nucleases, which along with Cas9 are widely used for targeted genome manipulation. Whereas Cas12 family nucleases are well characterized both biochemically and structurally, the molecular mechanism of TnpB remains unknown. Here we present the cryogenic-electron microscopy structures of the Deinococcus radiodurans TnpB-reRNA (right-end transposon element-derived RNA) complex in DNA-bound and -free forms. The structures reveal the basic architecture of TnpB nuclease and the molecular mechanism for DNA target recognition and cleavage that is supported by biochemical experiments. Collectively, these results demonstrate that TnpB represents the minimal structural and functional core of the Cas12 protein family and provide a framework for developing TnpB-based genome editing tools. |
 リンク リンク |  Nature / Nature /  PubMed:37020015 PubMed:37020015 |
| 手法 | EM (単粒子) |
| 解像度 | 2.8 - 3.14 Å |
| 構造データ | EMDB-16016, PDB-8bf8: EMDB-28655, PDB-8ex9: EMDB-28656, PDB-8exa: |
| 由来 |
|
 キーワード キーワード | RNA BINDING PROTEIN / Transposon / TnpB / reRNA / RuvC domain / Cas12 / IS200/IS605 |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について



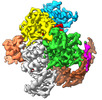



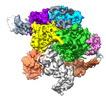

 Deinococcus radiodurans (放射線耐性)
Deinococcus radiodurans (放射線耐性)