+検索条件
-Structure paper
| タイトル | Cryo-EM structure of an active bacterial TIR-STING filament complex. |
|---|---|
| ジャーナル・号・ページ | Nature, Vol. 608, Issue 7924, Page 803-807, Year 2022 |
| 掲載日 | 2022年7月20日 |
 著者 著者 | Benjamin R Morehouse / Matthew C J Yip / Alexander F A Keszei / Nora K McNamara-Bordewick / Sichen Shao / Philip J Kranzusch /  |
| PubMed 要旨 | Stimulator of interferon genes (STING) is an antiviral signalling protein that is broadly conserved in both innate immunity in animals and phage defence in prokaryotes. Activation of STING requires ...Stimulator of interferon genes (STING) is an antiviral signalling protein that is broadly conserved in both innate immunity in animals and phage defence in prokaryotes. Activation of STING requires its assembly into an oligomeric filament structure through binding of a cyclic dinucleotide, but the molecular basis of STING filament assembly and extension remains unknown. Here we use cryogenic electron microscopy to determine the structure of the active Toll/interleukin-1 receptor (TIR)-STING filament complex from a Sphingobacterium faecium cyclic-oligonucleotide-based antiphage signalling system (CBASS) defence operon. Bacterial TIR-STING filament formation is driven by STING interfaces that become exposed on high-affinity recognition of the cognate cyclic dinucleotide signal c-di-GMP. Repeating dimeric STING units stack laterally head-to-head through surface interfaces, which are also essential for human STING tetramer formation and downstream immune signalling in mammals. The active bacterial TIR-STING structure reveals further cross-filament contacts that brace the assembly and coordinate packing of the associated TIR NADase effector domains at the base of the filament to drive NAD hydrolysis. STING interface and cross-filament contacts are essential for cell growth arrest in vivo and reveal a stepwise mechanism of activation whereby STING filament assembly is required for subsequent effector activation. Our results define the structural basis of STING filament formation in prokaryotic antiviral signalling. |
 リンク リンク |  Nature / Nature /  PubMed:35859168 / PubMed:35859168 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.3 - 4.3 Å |
| 構造データ | EMDB-26616, PDB-7un8: EMDB-26617, PDB-7un9: EMDB-26618, PDB-7una:  EMDB-26619: SfSTING with cGAMP |
| 化合物 | 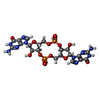 ChemComp-C2E: 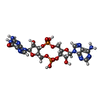 ChemComp-4BW: |
| 由来 |
|
 キーワード キーワード | ANTIVIRAL PROTEIN / STING / bacterial / filament |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について









 sphingobacterium faecium (バクテリア)
sphingobacterium faecium (バクテリア)