+検索条件
-Structure paper
| タイトル | Structure of the human cation-chloride cotransport KCC1 in an outward-open state. |
|---|---|
| ジャーナル・号・ページ | Proc Natl Acad Sci U S A, Vol. 119, Issue 27, Page e2109083119, Year 2022 |
| 掲載日 | 2022年7月5日 |
 著者 著者 | Yongxiang Zhao / Jiemin Shen / Qinzhe Wang / Manuel Jose Ruiz Munevar / Pietro Vidossich / Marco De Vivo / Ming Zhou / Erhu Cao /   |
| PubMed 要旨 | Cation-chloride cotransporters (CCCs) catalyze electroneutral symport of Cl with Na and/or K across membranes. CCCs are fundamental in cell volume homeostasis, transepithelia ion movement, ...Cation-chloride cotransporters (CCCs) catalyze electroneutral symport of Cl with Na and/or K across membranes. CCCs are fundamental in cell volume homeostasis, transepithelia ion movement, maintenance of intracellular Cl concentration, and neuronal excitability. Here, we present a cryoelectron microscopy structure of human K-Cl cotransporter (KCC)1 bound with the VU0463271 inhibitor in an outward-open state. In contrast to many other amino acid-polyamine-organocation transporter cousins, our first outward-open CCC structure reveals that opening the KCC1 extracellular ion permeation path does not involve hinge-bending motions of the transmembrane (TM) 1 and TM6 half-helices. Instead, rocking of TM3 and TM8, together with displacements of TM4, TM9, and a conserved intracellular loop 1 helix, underlie alternate opening and closing of extracellular and cytoplasmic vestibules. We show that KCC1 intriguingly exists in one of two distinct dimeric states via different intersubunit interfaces. Our studies provide a blueprint for understanding the mechanisms of CCCs and their inhibition by small molecule compounds. |
 リンク リンク |  Proc Natl Acad Sci U S A / Proc Natl Acad Sci U S A /  PubMed:35759661 / PubMed:35759661 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.25 - 3.5 Å |
| 構造データ | EMDB-26115, PDB-7tth: EMDB-26116, PDB-7tti: |
| 化合物 |  ChemComp-K: 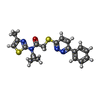 ChemComp-JUX: |
| 由来 |
|
 キーワード キーワード | TRANSPORT PROTEIN / SLC12A4 / Potassium-chloride transport / Inward-open state / TRANSPORT PROTEIN/INHIBITOR / VU0463271 / Outward-open state / TRANSPORT PROTEIN-INHIBITOR complex |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について







 homo sapiens (ヒト)
homo sapiens (ヒト)