+検索条件
-Structure paper
| タイトル | Cryo-EM structure of the mitochondrial protein-import channel TOM complex at near-atomic resolution. |
|---|---|
| ジャーナル・号・ページ | Nat Struct Mol Biol, Vol. 26, Issue 12, Page 1158-1166, Year 2019 |
| 掲載日 | 2019年11月18日 |
 著者 著者 | Kyle Tucker / Eunyong Park /  |
| PubMed 要旨 | Nearly all mitochondrial proteins are encoded by the nuclear genome and imported into mitochondria after synthesis on cytosolic ribosomes. These precursor proteins are translocated into mitochondria ...Nearly all mitochondrial proteins are encoded by the nuclear genome and imported into mitochondria after synthesis on cytosolic ribosomes. These precursor proteins are translocated into mitochondria by the TOM complex, a protein-conducting channel in the mitochondrial outer membrane. We have determined high-resolution cryo-EM structures of the core TOM complex from Saccharomyces cerevisiae in dimeric and tetrameric forms. Dimeric TOM consists of two copies each of five proteins arranged in two-fold symmetry: pore-forming β-barrel protein Tom40 and four auxiliary α-helical transmembrane proteins. The pore of each Tom40 has an overall negatively charged inner surface attributed to multiple functionally important acidic patches. The tetrameric complex is essentially a dimer of dimeric TOM, which may be capable of forming higher-order oligomers. Our study reveals the detailed molecular organization of the TOM complex and provides new insights about the mechanism of protein translocation into mitochondria. |
 リンク リンク |  Nat Struct Mol Biol / Nat Struct Mol Biol /  PubMed:31740857 / PubMed:31740857 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.06 - 4.1 Å |
| 構造データ | EMDB-20728, PDB-6ucu: EMDB-20729, PDB-6ucv: |
| 化合物 |  ChemComp-LMT: 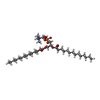 ChemComp-MC3: |
| 由来 |
|
 キーワード キーワード | TRANSLOCASE / Membrane protein / mitochondrial protein import / mitochondrial outer membrane / protein translocation |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について








