+検索条件
-Structure paper
| タイトル | Fidaxomicin jams RNA polymerase motions needed for initiation via RbpA contacts. |
|---|---|
| ジャーナル・号・ページ | Elife, Vol. 7, Year 2018 |
| 掲載日 | 2018年2月26日 |
 著者 著者 | Hande Boyaci / James Chen / Mirjana Lilic / Margaret Palka / Rachel Anne Mooney / Robert Landick / Seth A Darst / Elizabeth A Campbell /  |
| PubMed 要旨 | Fidaxomicin (Fdx) is an antimicrobial RNA polymerase (RNAP) inhibitor highly effective against RNAP in vitro, but clinical use of Fdx is limited to treating intestinal infections due to poor ...Fidaxomicin (Fdx) is an antimicrobial RNA polymerase (RNAP) inhibitor highly effective against RNAP in vitro, but clinical use of Fdx is limited to treating intestinal infections due to poor absorption. To identify the structural determinants of Fdx binding to RNAP, we determined the 3.4 Å cryo-electron microscopy structure of a complete RNAP holoenzyme in complex with Fdx. We find that the actinobacteria general transcription factor RbpA contacts fidaxomycin, explaining its strong effect on . Additional structures define conformational states of RNAP between the free apo-holoenzyme and the promoter-engaged open complex ready for transcription. The results establish that Fdx acts like a doorstop to jam the enzyme in an open state, preventing the motions necessary to secure promoter DNA in the active site. Our results provide a structural platform to guide development of anti-tuberculosis antimicrobials based on the Fdx binding pocket. |
 リンク リンク |  Elife / Elife /  PubMed:29480804 / PubMed:29480804 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.27 - 5.15 Å |
| 構造データ | |
| 化合物 | 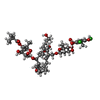 ChemComp-FI8:  ChemComp-ZN:  ChemComp-MG:  ChemComp-HOH: |
| 由来 |
|
 キーワード キーワード | transcription/dna/antibiotic / RNA Polymerase / Antibiotic / Inhibitor / TRANSCRIPTION / transcription-dna-antibiotic complex / transcription/dna / initiation / transcription bubble / closed clamp / transcription-dna complex / transcription/antibiotic / switch region inhibitor / transcription-antibiotic complex |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について












