+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-7322 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Mycobacterium tuberculosis RNAP Holo/RbpA in relaxed state | |||||||||
 Map data Map data | primary map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | initiation / transcription bubble / closed clamp / TRANSCRIPTION | |||||||||
| Function / homology |  Function and homology information Function and homology informationresponse to water / bacterial-type RNA polymerase holo enzyme binding / Antimicrobial action and antimicrobial resistance in Mtb / sigma factor antagonist complex / sigma factor activity / bacterial-type RNA polymerase core enzyme binding / cytosolic DNA-directed RNA polymerase complex / peptidoglycan-based cell wall / DNA-directed RNA polymerase complex / DNA-templated transcription initiation ...response to water / bacterial-type RNA polymerase holo enzyme binding / Antimicrobial action and antimicrobial resistance in Mtb / sigma factor antagonist complex / sigma factor activity / bacterial-type RNA polymerase core enzyme binding / cytosolic DNA-directed RNA polymerase complex / peptidoglycan-based cell wall / DNA-directed RNA polymerase complex / DNA-templated transcription initiation / ribonucleoside binding / DNA-directed RNA polymerase / DNA-directed RNA polymerase activity / nucleic acid binding / protein dimerization activity / transcription cis-regulatory region binding / response to antibiotic / regulation of DNA-templated transcription / DNA-templated transcription / positive regulation of DNA-templated transcription / magnesium ion binding / DNA binding / zinc ion binding / plasma membrane / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 5.15 Å | |||||||||
 Authors Authors | Darst SA / Campbell EA | |||||||||
 Citation Citation |  Journal: Elife / Year: 2018 Journal: Elife / Year: 2018Title: Fidaxomicin jams RNA polymerase motions needed for initiation via RbpA contacts. Authors: Hande Boyaci / James Chen / Mirjana Lilic / Margaret Palka / Rachel Anne Mooney / Robert Landick / Seth A Darst / Elizabeth A Campbell /  Abstract: Fidaxomicin (Fdx) is an antimicrobial RNA polymerase (RNAP) inhibitor highly effective against RNAP in vitro, but clinical use of Fdx is limited to treating intestinal infections due to poor ...Fidaxomicin (Fdx) is an antimicrobial RNA polymerase (RNAP) inhibitor highly effective against RNAP in vitro, but clinical use of Fdx is limited to treating intestinal infections due to poor absorption. To identify the structural determinants of Fdx binding to RNAP, we determined the 3.4 Å cryo-electron microscopy structure of a complete RNAP holoenzyme in complex with Fdx. We find that the actinobacteria general transcription factor RbpA contacts fidaxomycin, explaining its strong effect on . Additional structures define conformational states of RNAP between the free apo-holoenzyme and the promoter-engaged open complex ready for transcription. The results establish that Fdx acts like a doorstop to jam the enzyme in an open state, preventing the motions necessary to secure promoter DNA in the active site. Our results provide a structural platform to guide development of anti-tuberculosis antimicrobials based on the Fdx binding pocket. #1:  Journal: To Be Published Journal: To Be PublishedTitle: Structure of Mycobacterium Tuberculosis RNAP Holo Enzyme/RbpA in closed clamp conformation Authors: Darst SA / Campbell EA / Boyaci Selcuk H / Chen J / Lilic M | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_7322.map.gz emd_7322.map.gz | 37.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-7322-v30.xml emd-7322-v30.xml emd-7322.xml emd-7322.xml | 21.9 KB 21.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_7322.png emd_7322.png | 144.8 KB | ||
| Filedesc metadata |  emd-7322.cif.gz emd-7322.cif.gz | 8.7 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-7322 http://ftp.pdbj.org/pub/emdb/structures/EMD-7322 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7322 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7322 | HTTPS FTP |
-Related structure data
| Related structure data |  6c05MC  7319C  7320C  7323C  6bzoC  6c04C  6c06C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_7322.map.gz / Format: CCP4 / Size: 40.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_7322.map.gz / Format: CCP4 / Size: 40.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | primary map | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.5 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
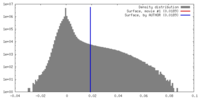
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Mycobacterium tuberculosis RNAP Holo with RbpA
| Entire | Name: Mycobacterium tuberculosis RNAP Holo with RbpA |
|---|---|
| Components |
|
-Supramolecule #1: Mycobacterium tuberculosis RNAP Holo with RbpA
| Supramolecule | Name: Mycobacterium tuberculosis RNAP Holo with RbpA / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#6 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: DNA-directed RNA polymerase subunit alpha
| Macromolecule | Name: DNA-directed RNA polymerase subunit alpha / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO / EC number: DNA-directed RNA polymerase |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 37.745328 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MLISQRPTLS EDVLTDNRSQ FVIEPLEPGF GYTLGNSLRR TLLSSIPGAA VTSIRIDGVL HEFTTVPGVK EDVTEIILNL KSLVVSSEE DEPVTMYLRK QGPGEVTAGD IVPPAGVTVH NPGMHIATLN DKGKLEVELV VERGRGYVPA VQNRASGAEI G RIPVDSIY ...String: MLISQRPTLS EDVLTDNRSQ FVIEPLEPGF GYTLGNSLRR TLLSSIPGAA VTSIRIDGVL HEFTTVPGVK EDVTEIILNL KSLVVSSEE DEPVTMYLRK QGPGEVTAGD IVPPAGVTVH NPGMHIATLN DKGKLEVELV VERGRGYVPA VQNRASGAEI G RIPVDSIY SPVLKVTYKV DATRVEQRTD FDKLILDVET KNSISPRDAL ASAGKTLVEL FGLARELNVE AEGIEIGPSP AE ADHIASF ALPIDDLDLT VRSYNCLKRE GVHTVGELVA RTESDLLDIR NFGQKSIDEV KIKLHQLGLS LKDSPPSFDP SEV AGYDVA TGTWSTEGAY DEQDYAETEQ L UniProtKB: DNA-directed RNA polymerase subunit alpha |
-Macromolecule #2: DNA-directed RNA polymerase subunit beta
| Macromolecule | Name: DNA-directed RNA polymerase subunit beta / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO / EC number: DNA-directed RNA polymerase |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 130.198922 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MADSRQSKTA ASPSPSRPQS SSNNSVPGAP NRVSFAKLRE PLEVPGLLDV QTDSFEWLIG SPRWRESAAE RGDVNPVGGL EEVLYELSP IEDFSGSMSL SFSDPRFDDV KAPVDECKDK DMTYAAPLFV TAEFINNNTG EIKSQTVFMG DFPMMTEKGT F IINGTERV ...String: MADSRQSKTA ASPSPSRPQS SSNNSVPGAP NRVSFAKLRE PLEVPGLLDV QTDSFEWLIG SPRWRESAAE RGDVNPVGGL EEVLYELSP IEDFSGSMSL SFSDPRFDDV KAPVDECKDK DMTYAAPLFV TAEFINNNTG EIKSQTVFMG DFPMMTEKGT F IINGTERV VVSQLVRSPG VYFDETIDKS TDKTLHSVKV IPSRGAWLEF DVDKRDTVGV RIDRKRRQPV TVLLKALGWT SE QIVERFG FSEIMRSTLE KDNTVGTDEA LLDIYRKLRP GEPPTKESAQ TLLENLFFKE KRYDLARVGR YKVNKKLGLH VGE PITSST LTEEDVVATI EYLVRLHEGQ TTMTVPGGVE VPVETDDIDH FGNRRLRTVG ELIQNQIRVG MSRMERVVRE RMTT QDVEA ITPQTLINIR PVVAAIKEFF GTSQLSQFMD QNNPLSGLTH KRRLSALGPG GLSRERAGLE VRDVHPSHYG RMCPI ETPE GPNIGLIGSL SVYARVNPFG FIETPYRKVV DGVVSDEIVY LTADEEDRHV VAQANSPIDA DGRFVEPRVL VRRKAG EVE YVPSSEVDYM DVSPRQMVSV ATAMIPFLEH DDANRALMGA NMQRQAVPLV RSEAPLVGTG MELRAAIDAG DVVVAEE SG VIEEVSADYI TVMHDNGTRR TYRMRKFARS NHGTCANQCP IVDAGDRVEA GQVIADGPCT DDGEMALGKN LLVAIMPW E GHNYEDAIIL SNRLVEEDVL TSIHIEEHEI DARDTKLGAE EITRDIPNIS DEVLADLDER GIVRIGAEVR DGDILVGKV TPKGETELTP EERLLRAIFG EKAREVRDTS LKVPHGESGK VIGIRVFSRE DEDELPAGVN ELVRVYVAQK RKISDGDKLA GRHGNKGVI GKILPVEDMP FLADGTPVDI ILNTHGVPRR MNIGQILETH LGWCAHSGWK VDAAKGVPDW AARLPDELLE A QPNAIVST PVFDGAQEAE LQGLLSCTLP NRDGDVLVDA DGKAMLFDGR SGEPFPYPVT VGYMYIMKLH HLVDDKIHAR ST GPYSMIT QQPLGGKAQF GGQRFGEMEC WAMQAYGAAY TLQELLTIKS DDTVGRVKVY EAIVKGENIP EPGIPESFKV LLK ELQSLC LNVEVLSSDG AAIELREGED EDLERAAANL GINLSRNESA SVEDLALARH GGSGA UniProtKB: DNA-directed RNA polymerase subunit beta |
-Macromolecule #3: DNA-directed RNA polymerase subunit beta'
| Macromolecule | Name: DNA-directed RNA polymerase subunit beta' / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO / EC number: DNA-directed RNA polymerase |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 148.074094 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MLDVNFFDEL RIGLATAEDI RQWSYGEVKK PETINYRTLK PEKDGLFCEK IFGPTRDWEC YCGKYKRVRF KGIICERCGV EVTRAKVRR ERMGHIELAA PVTHIWYFKG VPSRLGYLLD LAPKDLEKII YFAAYVITSV DEEMRHNELS TLEAEMAVER K AVEDQRDG ...String: MLDVNFFDEL RIGLATAEDI RQWSYGEVKK PETINYRTLK PEKDGLFCEK IFGPTRDWEC YCGKYKRVRF KGIICERCGV EVTRAKVRR ERMGHIELAA PVTHIWYFKG VPSRLGYLLD LAPKDLEKII YFAAYVITSV DEEMRHNELS TLEAEMAVER K AVEDQRDG ELEARAQKLE ADLAELEAEG AKADARRKVR DGGEREMRQI RDRAQRELDR LEDIWSTFTK LAPKQLIVDE NL YRELVDR YGEYFTGAMG AESIQKLIEN FDIDAEAESL RDVIRNGKGQ KKLRALKRLK VVAAFQQSGN SPMGMVLDAV PVI PPELRP MVQLDGGRFA TSDLNDLYRR VINRNNRLKR LIDLGAPEII VNNEKRMLQE SVDALFDNGR RGRPVTGPGN RPLK SLSDL LKGKQGRFRQ NLLGKRVDYS GRSVIVVGPQ LKLHQCGLPK LMALELFKPF VMKRLVDLNH AQNIKSAKRM VERQR PQVW DVLEEVIAEH PVLLNRAPTL HRLGIQAFEP MLVEGKAIQL HPLVCEAFNA DFDGDQMAVH LPLSAEAQAE ARILML SSN NILSPASGRP LAMPRLDMVT GLYYLTTEVP GDTGEYQPAS GDHPETGVYS SPAEAIMAAD RGVLSVRAKI KVRLTQL RP PVEIEAELFG HSGWQPGDAW MAETTLGRVM FNELLPLGYP FVNKQMHKKV QAAIINDLAE RYPMIVVAQT VDKLKDAG F YWATRSGVTV SMADVLVPPR KKEILDHYEE RADKVEKQFQ RGALNHDERN EALVEIWKEA TDEVGQALRE HYPDDNPII TIVDSGATGN FTQTRTLAGM KGLVTNPKGE FIPRPVKSSF REGLTVLEYF INTHGARKGL ADTALRTADS GYLTRRLVDV SQDVIVREH DCQTERGIVV ELAERAPDGT LIRDPYIETS AYARTLGTDA VDEAGNVIVE RGQDLGDPEI DALLAAGITQ V KVRSVLTC ATSTGVCATC YGRSMATGKL VDIGEAVGIV AAQSIGEPGT QLTMRTFHQG GVGEDITGGL PRVQELFEAR VP RGKAPIA DVTGRVRLED GERFYKITIV PDDGGEEVVY DKISKRQRLR VFKHEDGSER VLSDGDHVEV GQQLMEGSAD PHE VLRVQG PREVQIHLVR EVQEVYRAQG VSIHDKHIEV IVRQMLRRVT IIDSGSTEFL PGSLIDRAEF EAENRRVVAE GGEP AAGRP VLMGITKASL ATDSWLSAAS FQETTRVLTD AAINCRSDKL NGLKENVIIG KLIPAGTGIN RYRNIAVQPT EEARA AAYT IPSYEDQYYS PDFGAATGAA VPLDDYGYSD YRHHHHHHHH UniProtKB: DNA-directed RNA polymerase subunit beta' |
-Macromolecule #4: DNA-directed RNA polymerase subunit omega
| Macromolecule | Name: DNA-directed RNA polymerase subunit omega / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO / EC number: DNA-directed RNA polymerase |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 11.776996 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GSISQSDASL AAVPAVDQFD PSSGASGGYD TPLGITNPPI DELLDRVSSK YALVIYAAKR ARQINDYYNQ LGEGILEYVG PLVEPGLQE KPLSIALREI HADLLEHTEG E UniProtKB: DNA-directed RNA polymerase subunit omega |
-Macromolecule #5: RNA polymerase sigma factor SigA
| Macromolecule | Name: RNA polymerase sigma factor SigA / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 58.169477 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GPHMAATKAS TATDEPVKRT ATKSPAASAS GAKTGAKRTA AKSASGSPPA KRATKPAARS VKPASAPQDT TTSTIPKRKT RAAAKSAAA KAPSARGHAT KPRAPKDAQH EAATDPEDAL DSVEELDAEP DLDVEPGEDL DLDAADLNLD DLEDDVAPDA D DDLDSGDD ...String: GPHMAATKAS TATDEPVKRT ATKSPAASAS GAKTGAKRTA AKSASGSPPA KRATKPAARS VKPASAPQDT TTSTIPKRKT RAAAKSAAA KAPSARGHAT KPRAPKDAQH EAATDPEDAL DSVEELDAEP DLDVEPGEDL DLDAADLNLD DLEDDVAPDA D DDLDSGDD EDHEDLEAEA AVAPGQTADD DEEIAEPTEK DKASGDFVWD EDESEALRQA RKDAELTASA DSVRAYLKQI GK VALLNAE EEVELAKRIE AGLYATQLMT ELSERGEKLP AAQRRDMMWI CRDGDRAKNH LLEANLRLVV SLAKRYTGRG MAF LDLIQE GNLGLIRAVE KFDYTKGYKF STYATWWIRQ AITRAMADQA RTIRIPVHMV EVINKLGRIQ RELLQDLGRE PTPE ELAKE MDITPEKVLE IQQYAREPIS LDQTIGDEGD SQLGDFIEDS EAVVAVDAVS FTLLQDQLQS VLDTLSEREA GVVRL RFGL TDGQPRTLDE IGQVYGVTRE RIRQIESKTM SKLRHPSRSQ VLRDYLD UniProtKB: RNA polymerase sigma factor SigA |
-Macromolecule #6: RNA polymerase-binding protein RbpA
| Macromolecule | Name: RNA polymerase-binding protein RbpA / type: protein_or_peptide / ID: 6 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 12.993695 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MADRVLRGSR LGAVSYETDR NHDLAPRQIA RYRTDNGEEF EVPFADDAEI PGTWLCRNGM EGTLIEGDLP EPKKVKPPRT HWDMLLERR SIEELEELLK ERLELIRSRR RG UniProtKB: RNA polymerase-binding protein RbpA |
-Macromolecule #7: ZINC ION
| Macromolecule | Name: ZINC ION / type: ligand / ID: 7 / Number of copies: 2 / Formula: ZN |
|---|---|
| Molecular weight | Theoretical: 65.409 Da |
-Macromolecule #8: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 8 / Number of copies: 1 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 6.7 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller



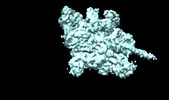











 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)