+検索条件
-Structure paper
| タイトル | Structure of human immunoproteasome with a reversible and noncompetitive inhibitor that selectively inhibits activated lymphocytes. |
|---|---|
| ジャーナル・号・ページ | Nat Commun, Vol. 8, Issue 1, Page 1692, Year 2017 |
| 掲載日 | 2017年11月22日 |
 著者 著者 | Ruda de Luna Almeida Santos / Lin Bai / Pradeep K Singh / Naoka Murakami / Hao Fan / Wenhu Zhan / Yingrong Zhu / Xiuju Jiang / Kaiming Zhang / Jean Pierre Assker / Carl F Nathan / Huilin Li / Jamil Azzi / Gang Lin /  |
| PubMed 要旨 | Proteasome inhibitors benefit patients with multiple myeloma and B cell-dependent autoimmune disorders but exert toxicity from inhibition of proteasomes in other cells. Toxicity should be minimized ...Proteasome inhibitors benefit patients with multiple myeloma and B cell-dependent autoimmune disorders but exert toxicity from inhibition of proteasomes in other cells. Toxicity should be minimized by reversible inhibition of the immunoproteasome β5i subunit while sparing the constitutive β5c subunit. Here we report β5i-selective inhibition by asparagine-ethylenediamine (AsnEDA)-based compounds and present the high-resolution cryo-EM structural analysis of the human immunoproteasome. Despite inhibiting noncompetitively, an AsnEDA inhibitor binds the active site. Hydrophobic interactions are accompanied by hydrogen bonding with β5i and β6 subunits. The inhibitors are far more cytotoxic for myeloma and lymphoma cell lines than for hepatocarcinoma or non-activated lymphocytes. They block human B-cell proliferation and promote apoptotic cell death selectively in antibody-secreting B cells, and to a lesser extent in activated human T cells. Reversible, β5i-selective inhibitors may be useful for treatment of diseases involving activated or neoplastic B cells or activated T cells. |
 リンク リンク |  Nat Commun / Nat Commun /  PubMed:29167449 / PubMed:29167449 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.8 Å |
| 構造データ | |
| 化合物 | 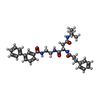 ChemComp-BZ7: |
| 由来 |
|
 キーワード キーワード | HYDROLASE/HYDROLASE INHIBITOR / inhibitor / complex / immunoproteasome / HYDROLASE-HYDROLASE INHIBITOR complex |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について





 homo sapiens (ヒト)
homo sapiens (ヒト)