+検索条件
-Structure paper
| タイトル | Structural and Functional Analyses of the Tridomain-Nonribosomal Peptide Synthetase FmoA3 for 4-Methyloxazoline Ring Formation. |
|---|---|
| ジャーナル・号・ページ | Angew Chem Int Ed Engl, Vol. 60, Issue 26, Page 14554-14562, Year 2021 |
| 掲載日 | 2021年6月21日 |
 著者 著者 | Yohei Katsuyama / Kaoru Sone / Ayaka Harada / Seiji Kawai / Naoki Urano / Naruhiko Adachi / Toshio Moriya / Masato Kawasaki / Kazuo Shin-Ya / Toshiya Senda / Yasuo Ohnishi /  |
| PubMed 要旨 | Nonribosomal peptide synthetases (NRPSs) are attractive targets for bioengineering to generate useful peptides. FmoA3 is a single modular NRPS composed of heterocyclization (Cy), adenylation (A), and ...Nonribosomal peptide synthetases (NRPSs) are attractive targets for bioengineering to generate useful peptides. FmoA3 is a single modular NRPS composed of heterocyclization (Cy), adenylation (A), and peptidyl carrier protein (PCP) domains. It uses α-methyl-l-serine to synthesize a 4-methyloxazoline ring, probably with another Cy domain in the preceding module FmoA2. Here, we determined the head-to-tail homodimeric structures of FmoA3 by X-ray crystallography (apo-form, with adenylyl-imidodiphosphate and α-methyl-l-seryl-AMP) and cryogenic electron microscopy single particle analysis, and performed site-directed mutagenesis experiments. The data revealed that α-methyl-l-serine can be accommodated in the active site because of the extra space around Ala688. The Cy domains of FmoA2 and FmoA3 catalyze peptide bond formation and heterocyclization, respectively. FmoA3's Cy domain seems to lose its donor PCP binding activity. The collective data support a proposed catalytic cycle of FmoA3. |
 リンク リンク |  Angew Chem Int Ed Engl / Angew Chem Int Ed Engl /  PubMed:33783097 PubMed:33783097 |
| 手法 | EM (単粒子) / X線回折 |
| 解像度 | 2.45 - 4.1 Å |
| 構造データ |  EMDB-30440:  PDB-6lta:  PDB-6ltb:  PDB-6ltc:  PDB-6ltd: |
| 化合物 | 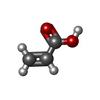 ChemComp-AKR:  ChemComp-HOH:  ChemComp-ANP:  ChemComp-AMP: 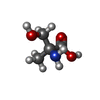 ChemComp-EW6:  ChemComp-SO4:  ChemComp-CL: |
| 由来 |
|
 キーワード キーワード | BIOSYNTHETIC PROTEIN / Nonribosomal peptide synthetases (NRPS) / JBIR-34 and -35 |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について



 streptomyces sp. sp080513ge-23 (バクテリア)
streptomyces sp. sp080513ge-23 (バクテリア)