+検索条件
-Structure paper
| タイトル | Structure and regulation of full-length human leucine-rich repeat kinase 1. |
|---|---|
| ジャーナル・号・ページ | Nat Commun, Vol. 14, Issue 1, Page 4797, Year 2023 |
| 掲載日 | 2023年8月9日 |
 著者 著者 | Riley D Metcalfe / Juliana A Martinez Fiesco / Luis Bonet-Ponce / Jillian H Kluss / Mark R Cookson / Ping Zhang /  |
| PubMed 要旨 | The human leucine-rich repeat kinases (LRRKs), LRRK1 and LRRK2 are large and unusually complex multi-domain kinases, which regulate fundamental cellular processes and have been implicated in human ...The human leucine-rich repeat kinases (LRRKs), LRRK1 and LRRK2 are large and unusually complex multi-domain kinases, which regulate fundamental cellular processes and have been implicated in human disease. Structures of LRRK2 have recently been determined, but the structure and molecular mechanisms regulating the activity of the LRRK1 as well as differences in the regulation of LRRK1 and LRRK2 remain unclear. Here, we report a cryo-EM structure of the LRRK1 monomer and a lower-resolution cryo-EM map of the LRRK1 dimer. The monomer structure, in which the kinase is in an inactive conformation, reveals key interdomain interfaces that control kinase activity as we validate experimentally. Both the LRRK1 monomer and dimer are structurally distinct compared to LRRK2. Overall, our results provide structural insights into the activation of the human LRRKs, which advance our understanding of their physiological and pathological roles. |
 リンク リンク |  Nat Commun / Nat Commun /  PubMed:37558661 / PubMed:37558661 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.78 - 6.38 Å |
| 構造データ | 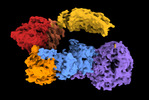 EMDB-28949: Leucine-rich repeat kinase 1 monomer, global refinement EMDB-28950, PDB-8fac: 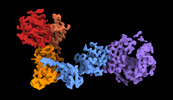 EMDB-28951: Leucine-rich repeat kinase 1 monomer, focused refinement on C-terminal region  EMDB-28952: Cryo-EM map of the LRRK1 dimer |
| 化合物 |  ChemComp-GDP: |
| 由来 |
|
 キーワード キーワード | TRANSFERASE / kinase / LRRK / multi-domain protein / GTPase |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について





 homo sapiens (ヒト)
homo sapiens (ヒト)