+検索条件
-Structure paper
| タイトル | Inhibition mechanism of NKCC1 involves the carboxyl terminus and long-range conformational coupling. |
|---|---|
| ジャーナル・号・ページ | Sci Adv, Vol. 8, Issue 43, Page eabq0952, Year 2022 |
| 掲載日 | 2022年10月28日 |
 著者 著者 | Mitchell A Moseng / Chih-Chia Su / Kerri Rios / Meng Cui / Meinan Lyu / Przemyslaw Glaza / Philip A Klenotic / Eric Delpire / Edward W Yu /  |
| PubMed 要旨 | The Na-K-2Cl cotransporter-1 (NKCC1) is an electroneutral Na-dependent transporter responsible for simultaneously translocating Na, K, and Cl ions into cells. In human tissue, NKCC1 plays a critical ...The Na-K-2Cl cotransporter-1 (NKCC1) is an electroneutral Na-dependent transporter responsible for simultaneously translocating Na, K, and Cl ions into cells. In human tissue, NKCC1 plays a critical role in regulating cytoplasmic volume, fluid intake, chloride homeostasis, and cell polarity. Here, we report four structures of human NKCC1 (hNKCC1), both in the absence and presence of loop diuretic (bumetanide or furosemide), using single-particle cryo-electron microscopy. These structures allow us to directly observe various novel conformations of the hNKCC1 dimer. They also reveal two drug-binding sites located at the transmembrane and cytosolic carboxyl-terminal domains, respectively. Together, our findings enable us to delineate an inhibition mechanism that involves a coupled movement between the cytosolic and transmembrane domains of hNKCC1. |
 リンク リンク |  Sci Adv / Sci Adv /  PubMed:36306358 / PubMed:36306358 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.28 - 3.87 Å |
| 構造データ | EMDB-24074, PDB-7mxo: EMDB-24141: Cryo-EM structure of human NKCC1 state Fu-I EMDB-25092: human NKCC1 state Fu-II EMDB-25204: Cryo-EM structure of NKCC1 Bu-I EMDB-40759, PDB-8ste: |
| 化合物 |  ChemComp-CL:  ChemComp-K: 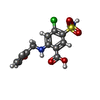 ChemComp-FUN: 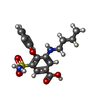 ChemComp-82U: |
| 由来 |
|
 キーワード キーワード | MEMBRANE PROTEIN / Sodium / potassium / chloride / co-transporter / ions / human / CTD / TMD / full-length / furosemide / diuretic / loop diuretic / NKCC1 / Transport Protein / cotransporter / dimer / ion |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について













 homo sapiens (ヒト)
homo sapiens (ヒト)