+検索条件
-Structure paper
| タイトル | Negative-Stain Electron Microscopy Reveals Dramatic Structural Rearrangements in Ni-Fe-S-Dependent Carbon Monoxide Dehydrogenase/Acetyl-CoA Synthase. |
|---|---|
| ジャーナル・号・ページ | Structure, Vol. 29, Issue 1, Page 43-49.e3, Year 2021 |
| 掲載日 | 2021年1月7日 |
 著者 著者 | Steven E Cohen / Edward J Brignole / Elizabeth C Wittenborn / Mehmet Can / Samuel Thompson / Stephen W Ragsdale / Catherine L Drennan /  |
| PubMed 要旨 | The Ni-Fe-S-containing A-cluster of acetyl-coenzyme A (CoA) synthase (ACS) assembles acetyl-CoA from carbon monoxide (CO), a methyl group (CH), and CoA. To accomplish this feat, ACS must bind CoA and ...The Ni-Fe-S-containing A-cluster of acetyl-coenzyme A (CoA) synthase (ACS) assembles acetyl-CoA from carbon monoxide (CO), a methyl group (CH), and CoA. To accomplish this feat, ACS must bind CoA and interact with two other proteins that contribute the CO and CH, respectively: CO dehydrogenase (CODH) and corrinoid Fe-S protein (CFeSP). Previous structural data show that, in the model acetogen Moorella thermoacetica, domain 1 of ACS binds to CODH such that a 70-Å-long internal channel is created that allows CO to travel from CODH to the A-cluster. The A-cluster is largely buried and is inaccessible to CFeSP for methylation. Here we use electron microscopy to capture multiple snapshots of ACS that reveal previously uncharacterized domain motion, forming extended and hyperextended structural states. In these structural states, the A-cluster is accessible for methylation by CFeSP. |
 リンク リンク |  Structure / Structure /  PubMed:32937101 / PubMed:32937101 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 44.8 - 46.7 Å |
| 構造データ |  EMDB-21005: 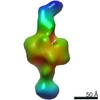 EMDB-21006:  EMDB-21007: 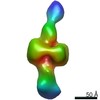 EMDB-21008: |
| 由来 |
|
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について



 Moorella thermoacetica (バクテリア)
Moorella thermoacetica (バクテリア)