+検索条件
-Structure paper
| タイトル | Cryo-EM structures reveal distinct mechanisms of inhibition of the human multidrug transporter ABCB1. |
|---|---|
| ジャーナル・号・ページ | Proc Natl Acad Sci U S A, Vol. 117, Issue 42, Page 26245-26253, Year 2020 |
| 掲載日 | 2020年10月20日 |
 著者 著者 | Kamil Nosol / Ksenija Romane / Rossitza N Irobalieva / Amer Alam / Julia Kowal / Naoya Fujita / Kaspar P Locher /    |
| PubMed 要旨 | ABCB1 detoxifies cells by exporting diverse xenobiotic compounds, thereby limiting drug disposition and contributing to multidrug resistance in cancer cells. Multiple small-molecule inhibitors and ...ABCB1 detoxifies cells by exporting diverse xenobiotic compounds, thereby limiting drug disposition and contributing to multidrug resistance in cancer cells. Multiple small-molecule inhibitors and inhibitory antibodies have been developed for therapeutic applications, but the structural basis of their activity is insufficiently understood. We determined cryo-EM structures of nanodisc-reconstituted, human ABCB1 in complex with the Fab fragment of the inhibitory, monoclonal antibody MRK16 and bound to a substrate (the antitumor drug vincristine) or to the potent inhibitors elacridar, tariquidar, or zosuquidar. We found that inhibitors bound in pairs, with one molecule lodged in the central drug-binding pocket and a second extending into a phenylalanine-rich cavity that we termed the "access tunnel." This finding explains how inhibitors can act as substrates at low concentration, but interfere with the early steps of the peristaltic extrusion mechanism at higher concentration. Our structural data will also help the development of more potent and selective ABCB1 inhibitors. |
 リンク リンク |  Proc Natl Acad Sci U S A / Proc Natl Acad Sci U S A /  PubMed:33020312 / PubMed:33020312 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.2 - 3.9 Å |
| 構造データ | EMDB-11666, PDB-7a65: EMDB-11667, PDB-7a69: EMDB-11670, PDB-7a6c: EMDB-11671, PDB-7a6e: EMDB-11672, PDB-7a6f: |
| 化合物 |  ChemComp-CLR: 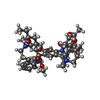 ChemComp-R1Q:  ChemComp-R0Z: 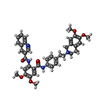 ChemComp-R1H: 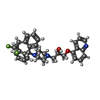 ChemComp-ZQU: |
| 由来 |
|
 キーワード キーワード | TRANSPORT PROTEIN / P-glycoprotein / MDR1 / nanodisc |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について













 homo sapiens (ヒト)
homo sapiens (ヒト)
