+検索条件
-Structure paper
| タイトル | Activation and Signaling Mechanism Revealed by Cannabinoid Receptor-G Complex Structures. |
|---|---|
| ジャーナル・号・ページ | Cell, Vol. 180, Issue 4, Page 655-665.e18, Year 2020 |
| 掲載日 | 2020年2月20日 |
 著者 著者 | Tian Hua / Xiaoting Li / Lijie Wu / Christos Iliopoulos-Tsoutsouvas / Yuxia Wang / Meng Wu / Ling Shen / Christina A Brust / Spyros P Nikas / Feng Song / Xiyong Song / Shuguang Yuan / Qianqian Sun / Yiran Wu / Shan Jiang / Travis W Grim / Othman Benchama / Edward L Stahl / Nikolai Zvonok / Suwen Zhao / Laura M Bohn / Alexandros Makriyannis / Zhi-Jie Liu /   |
| PubMed 要旨 | Human endocannabinoid systems modulate multiple physiological processes mainly through the activation of cannabinoid receptors CB1 and CB2. Their high sequence similarity, low agonist selectivity, ...Human endocannabinoid systems modulate multiple physiological processes mainly through the activation of cannabinoid receptors CB1 and CB2. Their high sequence similarity, low agonist selectivity, and lack of activation and G protein-coupling knowledge have hindered the development of therapeutic applications. Importantly, missing structural information has significantly held back the development of promising CB2-selective agonist drugs for treating inflammatory and neuropathic pain without the psychoactivity of CB1. Here, we report the cryoelectron microscopy structures of synthetic cannabinoid-bound CB2 and CB1 in complex with G, as well as agonist-bound CB2 crystal structure. Of important scientific and therapeutic benefit, our results reveal a diverse activation and signaling mechanism, the structural basis of CB2-selective agonists design, and the unexpected interaction of cholesterol with CB1, suggestive of its endogenous allosteric modulating role. |
 リンク リンク |  Cell / Cell /  PubMed:32004463 / PubMed:32004463 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) / X線回折 |
| 解像度 | 2.9 - 3.2 Å |
| 構造データ | EMDB-0744, PDB-6kpf:  PDB-6kpc: |
| 化合物 | 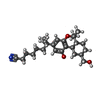 ChemComp-E3R: 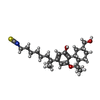 ChemComp-8D0: |
| 由来 |
|
 キーワード キーワード | MEMBRANE PROTEIN / GPCR / LCP / agonist / Gi Protein / G protein / cryo-EM |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について







 homo sapiens (ヒト)
homo sapiens (ヒト) enterobacteria phage rb59 (ファージ)
enterobacteria phage rb59 (ファージ)