+検索条件
-Structure paper
| タイトル | The domain architecture of the protozoan protein J-DNA-binding protein 1 suggests synergy between base J DNA binding and thymidine hydroxylase activity. |
|---|---|
| ジャーナル・号・ページ | J Biol Chem, Vol. 294, Issue 34, Page 12815-12825, Year 2019 |
| 掲載日 | 2019年8月23日 |
 著者 著者 | Athanassios Adamopoulos / Tatjana Heidebrecht / Jeroen Roosendaal / Wouter G Touw / Isabelle Q Phan / Jos Beijnen / Anastassis Perrakis /   |
| PubMed 要旨 | J-DNA-binding protein 1 (JBP1) contributes to the biosynthesis and maintenance of base J (β-d-glucosyl-hydroxymethyluracil), an epigenetic modification of thymidine (T) confined to pathogenic ...J-DNA-binding protein 1 (JBP1) contributes to the biosynthesis and maintenance of base J (β-d-glucosyl-hydroxymethyluracil), an epigenetic modification of thymidine (T) confined to pathogenic protozoa such as and JBP1 has two known functional domains: an N-terminal T hydroxylase (TH) homologous to the 5-methylcytosine hydroxylase domain in TET proteins and a J-DNA-binding domain (JDBD) that resides in the middle of JBP1. Here, we show that removing JDBD from JBP1 results in a soluble protein (Δ-JDBD) with the N- and C-terminal regions tightly associated together in a well-ordered structure. We found that this Δ-JDBD domain retains TH activity but displays a 15-fold lower apparent rate of hydroxylation compared with JBP1. Small-angle X-ray scattering (SAXS) experiments on JBP1 and JDBD in the presence or absence of J-DNA and on Δ-JDBD enabled us to generate low-resolution three-dimensional models. We conclude that Δ-JDBD, and not the N-terminal region of JBP1 alone, is a distinct folding unit. Our SAXS-based model supports the notion that binding of JDBD specifically to J-DNA can facilitate T hydroxylation 12-14 bp downstream on the complementary strand of the J-recognition site. We postulate that insertion of the JDBD module into the Δ-JDBD scaffold during evolution provided a mechanism that synergized J recognition and T hydroxylation, ensuring inheritance of base J in specific sequence patterns following DNA replication in kinetoplastid parasites. |
 リンク リンク |  J Biol Chem / J Biol Chem /  PubMed:31292194 / PubMed:31292194 /  PubMed Central PubMed Central |
| 手法 | SAS (X-ray synchrotron) |
| 構造データ | 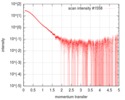 SASDGP2: Thymine dioxygenase full length J-DNA binding protein (JBP1) 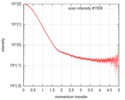 SASDGQ2: Thymine dioxygenase lacking the JDNA binding domain Delta-JDBD (Δ-JDBD) 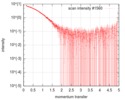 SASDGR2: Thymine dioxygenase J-containing DNA binding domain (JDBD) 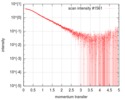 SASDGS2: J-23-DNA (J-DNA (23mer)) 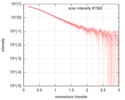 SASDGT2: Thymine dioxygenase J-containing DNA binding domain in complex with J-23-DNA (JDBD:J-23-DNA) 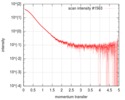 SASDGU2: Thymine dioxygenase full length J-DNA binding protein in complex with J-23-DNA (JBP1:J-23-DNA) |
| 由来 |
|
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について



 Leishmania tarentolae (真核生物)
Leishmania tarentolae (真核生物)