+Search query
-Structure paper
| Title | Structural basis for the oligomerization-mediated regulation of NLRP3 inflammasome activation. |
|---|---|
| Journal, issue, pages | Proc Natl Acad Sci U S A, Vol. 119, Issue 11, Page e2121353119, Year 2022 |
| Publish date | Mar 15, 2022 |
 Authors Authors | Umeharu Ohto / Yukie Kamitsukasa / Hanako Ishida / Zhikuan Zhang / Karin Murakami / Chie Hirama / Sakiko Maekawa / Toshiyuki Shimizu /  |
| PubMed Abstract | SignificanceThe nucleotide-binding oligomerization domain (NOD)-like receptor pyrin domain containing 3 (NLRP3) is a pattern recognition receptor that forms an inflammasome. The cryo-electron ...SignificanceThe nucleotide-binding oligomerization domain (NOD)-like receptor pyrin domain containing 3 (NLRP3) is a pattern recognition receptor that forms an inflammasome. The cryo-electron microscopy structure of the dodecameric form of full-length NLRP3 bound to the clinically relevant NLRP3-specific inhibitor MCC950 has established the structural basis for the oligomerization-mediated regulation of NLRP3 inflammasome activation and the mechanism of action of the NLRP3 specific inhibitor. The inactive NLRP3 oligomer represents the NLRP3 resting state, capable of binding to membranes and is likely disrupted for its activation. Visualization of the inhibitor binding mode will enable optimization of the activity of NLRP3 inflammasome inhibitor drugs. |
 External links External links |  Proc Natl Acad Sci U S A / Proc Natl Acad Sci U S A /  PubMed:35254907 / PubMed:35254907 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 3.23 - 3.55 Å |
| Structure data | EMDB-32119, PDB-7vtp: EMDB-32120, PDB-7vtq: |
| Chemicals |  ChemComp-ADP: 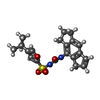 ChemComp-7YN: |
| Source |
|
 Keywords Keywords | IMMUNE SYSTEM / NLR / NOD-like receptor / NLRP3 / Inflammasome |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers







 homo sapiens (human)
homo sapiens (human)
