+Search query
-Structure paper
| Title | Immunodominant proteins P1 and P40/P90 from human pathogen Mycoplasma pneumoniae. |
|---|---|
| Journal, issue, pages | Nat Commun, Vol. 11, Issue 1, Page 5188, Year 2020 |
| Publish date | Oct 14, 2020 |
 Authors Authors | David Vizarraga / Akihiro Kawamoto / U Matsumoto / Ramiro Illanes / Rosa Pérez-Luque / Jesús Martín / Rocco Mazzolini / Paula Bierge / Oscar Q Pich / Mateu Espasa / Isabel Sanfeliu / Juliana Esperalba / Miguel Fernández-Huerta / Margot P Scheffer / Jaume Pinyol / Achilleas S Frangakis / Maria Lluch-Senar / Shigetarou Mori / Keigo Shibayama / Tsuyoshi Kenri / Takayuki Kato / Keiichi Namba / Ignacio Fita / Makoto Miyata / David Aparicio /    |
| PubMed Abstract | Mycoplasma pneumoniae is a bacterial human pathogen that causes primary atypical pneumonia. M. pneumoniae motility and infectivity are mediated by the immunodominant proteins P1 and P40/P90, which ...Mycoplasma pneumoniae is a bacterial human pathogen that causes primary atypical pneumonia. M. pneumoniae motility and infectivity are mediated by the immunodominant proteins P1 and P40/P90, which form a transmembrane adhesion complex. Here we report the structure of P1, determined by X-ray crystallography and cryo-electron microscopy, and the X-ray structure of P40/P90. Contrary to what had been suggested, the binding site for sialic acid was found in P40/P90 and not in P1. Genetic and clinical variability concentrates on the N-terminal domain surfaces of P1 and P40/P90. Polyclonal antibodies generated against the mostly conserved C-terminal domain of P1 inhibited adhesion of M. pneumoniae, and serology assays with sera from infected patients were positive when tested against this C-terminal domain. P40/P90 also showed strong reactivity against human infected sera. The architectural elements determined for P1 and P40/P90 open new possibilities in vaccine development against M. pneumoniae infections. |
 External links External links |  Nat Commun / Nat Commun /  PubMed:33057023 / PubMed:33057023 /  PubMed Central PubMed Central |
| Methods | EM (single particle) / X-ray diffraction |
| Resolution | 1.94 - 3.1 Å |
| Structure data | EMDB-30233, PDB-7bwm:  PDB-6rc9:  PDB-6rj1:  PDB-6tlz:  PDB-6tm0: |
| Chemicals |  ChemComp-HOH: 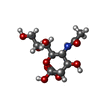 ChemComp-SIA: |
| Source |
|
 Keywords Keywords | CELL ADHESION / adhesin / extracellular / transmembrane-complex / immunodominant protein. / Adhesion / Sugars / STRUCTURAL PROTEIN / Sialic acid receptor |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers





 mycoplasma pneumoniae m129 (bacteria)
mycoplasma pneumoniae m129 (bacteria)