+Search query
-Structure paper
| Title | Structural basis for the inhibition mechanism of the DNA polymerase holoenzyme from mpox virus. |
|---|---|
| Journal, issue, pages | Structure, Vol. 32, Issue 6, Page 654-661.e3, Year 2024 |
| Publish date | Jun 6, 2024 |
 Authors Authors | Yaping Shen / Yaning Li / Renhong Yan /  |
| PubMed Abstract | There are three key components at the core of the mpox virus (MPXV) DNA polymerase holoenzyme: DNA polymerase F8, processivity factors A22, and the Uracil-DNA glycosylase E4. The holoenzyme is ...There are three key components at the core of the mpox virus (MPXV) DNA polymerase holoenzyme: DNA polymerase F8, processivity factors A22, and the Uracil-DNA glycosylase E4. The holoenzyme is recognized as a vital antiviral target because MPXV replicates in the cytoplasm of host cells. Nucleotide analogs such as cidofovir and cytarabine (Ara-C) have shown potential in curbing MPXV replication and they also display promise against other poxviruses. However, the mechanism behind their inhibitory effects remains unclear. Here, we present the cryo-EM structure of the DNA polymerase holoenzyme F8/A22/E4 bound with its competitive inhibitor Ara-C-derived cytarabine triphosphate (Ara-CTP) at an overall resolution of 3.0 Å and reveal its inhibition mechanism. Ara-CTP functions as a direct chain terminator in proximity to the deoxycytidine triphosphate (dCTP)-binding site. The extra hydrogen bond formed with Asn665 makes it more potent in binding than dCTP. Asn665 is conserved among eukaryotic B-family polymerases. |
 External links External links |  Structure / Structure /  PubMed:38579705 PubMed:38579705 |
| Methods | EM (single particle) |
| Resolution | 3.0 - 3.06 Å |
| Structure data | EMDB-36960, PDB-8k8s: EMDB-36962, PDB-8k8u:  EMDB-36963: the local map of DNA and Ara-CTP binding site  EMDB-36964: the local map of DNA and dCTP binding site |
| Chemicals |  ChemComp-MG:  ChemComp-HF4:  ChemComp-HOH: 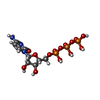 ChemComp-CTP: |
| Source |
|
 Keywords Keywords | VIRAL PROTEIN/DNA / RECOMBINATION / REPLICATION / VIRAL PROTEIN-DNA COMPLEX |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers







 monkeypox virus
monkeypox virus