+Search query
-Structure paper
| Title | Structural basis for inhibition of the SARS-CoV-2 RNA polymerase by suramin. |
|---|---|
| Journal, issue, pages | Nat Struct Mol Biol, Vol. 28, Issue 3, Page 319-325, Year 2021 |
| Publish date | Mar 5, 2021 |
 Authors Authors | Wanchao Yin / Xiaodong Luan / Zhihai Li / Ziwei Zhou / Qingxing Wang / Minqi Gao / Xiaoxi Wang / Fulai Zhou / Jingjing Shi / Erli You / Mingliang Liu / Qingxia Wang / Yi Jiang / Hualiang Jiang / Gengfu Xiao / Leike Zhang / Xuekui Yu / Shuyang Zhang / H Eric Xu /  |
| PubMed Abstract | The COVID-19 pandemic caused by nonstop infections of SARS-CoV-2 has continued to ravage many countries worldwide. Here we report that suramin, a 100-year-old drug, is a potent inhibitor of the SARS- ...The COVID-19 pandemic caused by nonstop infections of SARS-CoV-2 has continued to ravage many countries worldwide. Here we report that suramin, a 100-year-old drug, is a potent inhibitor of the SARS-CoV-2 RNA-dependent RNA polymerase (RdRp) and acts by blocking the binding of RNA to the enzyme. In biochemical assays, suramin and its derivatives are at least 20-fold more potent than remdesivir, the currently approved nucleotide drug for treatment of COVID-19. The 2.6 Å cryo-electron microscopy structure of the viral RdRp bound to suramin reveals two binding sites. One site directly blocks the binding of the RNA template strand and the other site clashes with the RNA primer strand near the RdRp catalytic site, thus inhibiting RdRp activity. Suramin blocks viral replication in Vero E6 cells, although the reasons underlying this effect are likely various. Our results provide a structural mechanism for a nonnucleotide inhibitor of the SARS-CoV-2 RdRp. |
 External links External links |  Nat Struct Mol Biol / Nat Struct Mol Biol /  PubMed:33674802 PubMed:33674802 |
| Methods | EM (single particle) |
| Resolution | 2.57 Å |
| Structure data | EMDB-30572, PDB-7d4f: |
| Chemicals |  ChemComp-ZN: 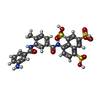 ChemComp-H3U: |
| Source |
|
 Keywords Keywords | VIRAL PROTEIN / COVID-19 / RNA polymerase / suramin binding |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers






