+Search query
-Structure paper
| Title | DOLORS: versatile strategy for internal labeling and domain localization in electron microscopy. |
|---|---|
| Journal, issue, pages | Structure, Vol. 20, Issue 12, Page 1995-2002, Year 2012 |
| Publish date | Dec 5, 2012 |
 Authors Authors | Pick-Wei Lau / Clinton S Potter / Bridget Carragher / Ian J MacRae /  |
| PubMed Abstract | Single-particle electron microscopy (EM) is a powerful tool for studying the structures of large biological molecules. However, the achievable resolution does not always allow for direct recognition ...Single-particle electron microscopy (EM) is a powerful tool for studying the structures of large biological molecules. However, the achievable resolution does not always allow for direct recognition of individual protein domains. Labels that can be visualized by EM have been developed for protein termini, but tagging internal domains remains a challenge. We describe a robust strategy for determining the position of internal sites within EM maps, termed domain localization by RCT sampling (DOLORS). DOLORS uses monovalent streptavidin added posttranslationally to tagged sites in the target protein. Internal labels generally display less conformational flexibility than terminal labels, providing more precise positional information. Automated methods are used to rapidly generate assemblies of unique 3D models allowing the attachment sites of labeled domains to be accurately identified and thus provide an overall architectural map of the molecule. |
 External links External links |  Structure / Structure /  PubMed:23217681 / PubMed:23217681 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Structure data |  EMDB-2214: 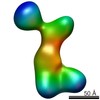 EMDB-2215: 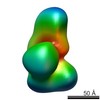 EMDB-2216:  EMDB-2217:  EMDB-2218: |
| Source |
|
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers



 Homo sapiens (human)
Homo sapiens (human) Streptomyces avidinii (bacteria)
Streptomyces avidinii (bacteria)