+検索条件
-Structure paper
| タイトル | N-type fast inactivation of a eukaryotic voltage-gated sodium channel. |
|---|---|
| ジャーナル・号・ページ | Nat Commun, Vol. 13, Issue 1, Page 2713, Year 2022 |
| 掲載日 | 2022年5月17日 |
 著者 著者 | Jiangtao Zhang / Yiqiang Shi / Junping Fan / Huiwen Chen / Zhanyi Xia / Bo Huang / Juquan Jiang / Jianke Gong / Zhuo Huang / Daohua Jiang /  |
| PubMed 要旨 | Voltage-gated sodium (Na) channels initiate action potentials. Fast inactivation of Na channels, mediated by an Ile-Phe-Met motif, is crucial for preventing hyperexcitability and regulating firing ...Voltage-gated sodium (Na) channels initiate action potentials. Fast inactivation of Na channels, mediated by an Ile-Phe-Met motif, is crucial for preventing hyperexcitability and regulating firing frequency. Here we present cryo-electron microscopy structure of NaEh from the coccolithophore Emiliania huxleyi, which reveals an unexpected molecular gating mechanism for Na channel fast inactivation independent of the Ile-Phe-Met motif. An N-terminal helix of NaEh plugs into the open activation gate and blocks it. The binding pose of the helix is stabilized by multiple electrostatic interactions. Deletion of the helix or mutations blocking the electrostatic interactions completely abolished the fast inactivation. These strong interactions enable rapid inactivation, but also delay recovery from fast inactivation, which is ~160-fold slower than human Na channels. Together, our results provide mechanistic insights into fast inactivation of NaEh that fundamentally differs from the conventional local allosteric inhibition, revealing both surprising structural diversity and functional conservation of ion channel inactivation. |
 リンク リンク |  Nat Commun / Nat Commun /  PubMed:35581266 / PubMed:35581266 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 2.83 Å |
| 構造データ | EMDB-33016: Ion channel |
| 由来 |
|
 キーワード キーワード | TRANSPORT PROTEIN / ion channel |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について



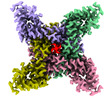

 emiliania huxleyi (真核生物)
emiliania huxleyi (真核生物)