+Search query
-Structure paper
| Title | Molecular basis for N-terminal alpha-synuclein acetylation by human NatB. |
|---|---|
| Journal, issue, pages | Elife, Vol. 9, Year 2020 |
| Publish date | Sep 4, 2020 |
 Authors Authors | Sunbin Deng / Buyan Pan / Leah Gottlieb / E James Petersson / Ronen Marmorstein /  |
| PubMed Abstract | NatB is one of three major N-terminal acetyltransferase (NAT) complexes (NatA-NatC), which co-translationally acetylate the N-termini of eukaryotic proteins. Its substrates account for about 21% of ...NatB is one of three major N-terminal acetyltransferase (NAT) complexes (NatA-NatC), which co-translationally acetylate the N-termini of eukaryotic proteins. Its substrates account for about 21% of the human proteome, including well known proteins such as actin, tropomyosin, CDK2, and α-synuclein (αSyn). Human NatB (hNatB) mediated N-terminal acetylation of αSyn has been demonstrated to play key roles in the pathogenesis of Parkinson's disease and as a potential therapeutic target for hepatocellular carcinoma. Here we report the cryo-EM structure of hNatB bound to a CoA-αSyn conjugate, together with structure-guided analysis of mutational effects on catalysis. This analysis reveals functionally important differences with human NatA and NatB, resolves key hNatB protein determinants for αSyn N-terminal acetylation, and identifies important residues for substrate-specific recognition and acetylation by NatB enzymes. These studies have implications for developing small molecule NatB probes and for understanding the mode of substrate selection by NAT enzymes. |
 External links External links |  Elife / Elife /  PubMed:32885784 / PubMed:32885784 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 3.46 Å |
| Structure data | EMDB-21307, PDB-6vp9: |
| Chemicals | 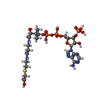 ChemComp-CMC: |
| Source |
|
 Keywords Keywords | TRANSFERASE / NatB / NAA20 / NAA25 |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers





 homo sapiens (human)
homo sapiens (human)
